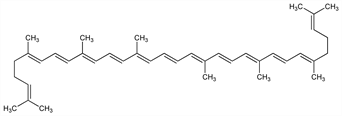
5620. Lycopene
Nomenclature
Description and references
Carotenoid antioxidant occurring in ripe fruit, especially in tomatoes. One kg of fresh ripe tomatoes yields 0.02 g lycopene. Isoln procedure: R. Willst"atter, H. H. Escher, Z. Physiol. Chem. 64, 47 (1910). Chromatographic sepn from other carotenoids: A. Winterstein, ibid. 215, 51 (1933); A. Winterstein, G. Stein, ibid. 220, 247 (1933). HPLC determn in plasma: M. V. Vertzoni et al., J. Chromatogr. B 819, 149 (2005). Structure: Willst"atter, Escher, loc. cit.; Karrer and collaborators: Helv. Chim. Acta 11, 751, 1201 (1928); 12, 285 (1929); 13, 1084 (1930); 14, 435 (1931); Kuhn, Grundmann, Ber. 65, 898, 1880 (1932). Synthesis: P. Karrer et al., Helv. Chim. Acta 33, 1349 (1950); O. Isler et al., ibid. 39, 463 (1956). Commercial prepn: Kabbe et al., DE 1168890 (1964 to Bayer). Oral toxicity study: W. Mellert et al., Food Chem. Toxicol. 40, 1581 (2002). Review of antioxidant activity and role in human health: A. V. Rao, S. Agarwal, Nutr. Res. 19, 305-323 (1999).