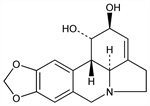
5626. Lycorine
Nomenclature
Description and references
Alkaloid isolated from the bulbs of Lycoris radiata L., Narcissus pseudonarcissus L., N. tazetta L., from Buphane disticha Herb., in Crinum spp., Amaryllis belladonna L., Clivia miniata Regel and other Amaryllidaceae. Extraction procedure: Cook, Loudon in Manske-Holmes, Alkaloids vol. II (Academic Press, 1952) p 336. Identity of lycorine and galanthidine: Proskurnina, Dokl. Akad. Nauk SSSR 90, 565 (1953), C.A. 49, 12500c (1955). Structure: Takeda et al., J. Am. Chem. Soc. 80, 2562 (1958). Stereochemistry: Nakagawa, Uyeo, J. Chem. Soc. 1959, 3736; K. Kotera et al., Tetrahedron Lett. 1966, 2009. Crystal structure: R. Roques, Acta Crystallogr. 30B, 296 (1974). Attempted synthesis: Dyke et al., Tetrahedron 29, 213 (1973). Synthesis: Y. Tsuda et al., Chem. Commun. 1975, 933; eidem, J. Chem. Soc. Perkin Trans. 1 1979, 1358; T. Sano et al., Heterocycles 14, 1097 (1980); S. F. Martin, C. Tu, J. Org. Chem. 46, 3763 (1981). Biosynthesis: Archer et al., Proc. Chem. Soc. London 1963, 168; Fugati, Mazza, Chem. Commun. 1972, 936. Review: W. C. Wildman in The Alkaloids vol. XI, R. H. F. Manske, Ed. (Academic Press, New York, 1968) pp 307-400.