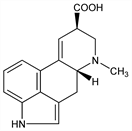
5633. Lysergic Acid
Nomenclature
Description and references
Lysergic acid and isolysergic acid are the main cleavage products formed on alkaline hydrolysis of the alkaloids which are characteristic of ergot. Jacobs, Craig et al., J. Biol. Chem. 104, 547 (1934); 125, 289 (1938); 130, 399 (1939); 145, 487 (1942); J. Org. Chem. 10, 76 (1945). High-yield production by Claviceps paspali: Arcamone et al., Proc. R. Soc. London Ser. B 155, 26 (1961). Total synthesis: Kornfeld et al., J. Am. Chem. Soc. 76, 5256 (1954); 78, 3087 (1956); M. Julia et al., Tetrahedron Lett. 1969, 1569; V. W. Armstrong et al., ibid. 1976, 4311; W. Oppolzer et al., Helv. Chim. Acta 64, 478 (1981); R. Ramage et al., Tetrahedron 37, Suppl. 9, 157 (1981); J. Rebek, D. F. Tai, Tetrahedron Lett. 24, 859 (1983). Stereochemistry: Stoll et al., Helv. Chim. Acta 37, 2039 (1954); Stenlake, J. Chem. Soc. 1955, 1626; Leeman, Fabbri, Helv. Chim. Acta 42, 2696 (1959). Absolute configuration: Stadler, Hofmann, ibid. 45, 2005 (1962).