5634. Lysergide
Nomenclature
CAS number: 50-37-3
9,10-Didehydro-N,N-diethyl-6-methylergoline-8β-carboxamide; N,N-diethyl-d-lysergamide; d-lysergic acid diethylamide; LSD; LSD-25; lysergs"aure diethylamid. C20H25N3O; mol wt 323.43.
C 74.27%, H 7.79%, N 12.99%, O 4.95%.
Description and references
Microbial formation by Claviceps paspali over the hydroxyethylamide: Arcamone et al., Proc. R. Soc. London 155B, 26 (1961). Partial synthesis: Stoll, Hofmann, Helv. Chim. Acta 26, 944 (1943); 38, 421 (1955). Industrial prepn: Pioch, US 2736728; Garbrecht, US 2774763 (both 1956 to Lilly); Patelli, Bernardi, US 3141887 (1964 to Farmitalia). Isotope-labeled LSD: Stoll et al., Helv. Chim. Acta 37, 820 (1954). LC-MS determn in biological fluids: J. Canezin et al., J. Chromatogr. B 765, 15 (2001). Toxicity data: E. Rothlin, Ann. N.Y. Acad. Sci. 66, 668 (1957). Review: Hoffer, Clin. Pharmacol. Ther. 6, 183 (1965). Book: The Use of LSD in Psychotherapy and Alcoholism, H. A. Abramson, Ed. (Bobbs-Merrill, Indianapolis, 1967) 697 pp.
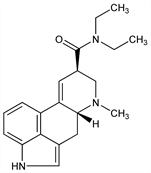
Properties
Pointed prisms from benzene, mp 80-85°. [α]D20 +17° (c = 0.5 in pyridine). uv max (ethanol): 311 nm (E1%1cm 257). LD50 in mice, rats, rabbits (mg/kg): 46, 16.5, 0.3 i.v. (Rothlin).Derivative
d-Tartrate.C46H64N6O10; mol wt 861.03.
C 64.17%, H 7.49%, N 9.76%, O 18.58%.