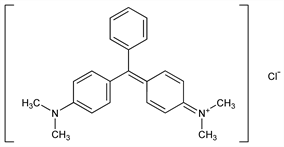
Nomenclature
CAS number: 569-64-2;
510-13-4
(carbinol base)
N-[4-[[4-(Dimethylamino)phenyl]phenylmethylene]-2,5-cyclohexadien-1-ylidene]-
N-methylmethanaminium chloride; bis[
p-(dimethylamino)phenyl]phenylmethylium
chloride; C.I. Basic Green 4; C.I. 42000; aniline green; benzal green; benzaldehyde green; china green; diamond green B; diamond green Bx; diamond green P extra; fast green; light green N; new victoria green extra O; new victoria green extra I; new victoria green extra II; solid green O; victoria green W; victoria green B.
C
23H
25ClN
2; mol wt 364.91.
C 75.70%, H 6.91%, Cl 9.72%, N 7.68%.
Description and references
Triphenylmethane dye with fungicidal and limited
antiseptic activity. Prepn: Colour Index Vol. 4 (3rd ed., 1971) p 4380. Conformational changes
in excited state: S. Saikan, J. Sei, J. Chem.
Phys. 79, 4154 (1983). Use in electron microscopy:
R. G. Pourcho et al., Stain Technol. 53, 29 (1978); as stain in determn of prostaglandin E
and F compounds: E. J. Singh et al., J. Chromatogr. 105, 195 (1975); in determn
of inorganic phosphate: W. Hohenwallner, E. Wimmer, Clin. Chim. Acta 45, 169 (1973);
S. G. Carter, D. W. Karl, J. Biochem. Biophys.
Methods 7, 7 (1982). Antiseptic activity:
J. E. Madden et al., Surg. Forum 22, 63 (1971). Toxicology: N. Brock, A. Erhardt, Arzneim.-Forsch. 1, 5 (1951);
T. D. Bills, L. L. Marking, Invest. Fish Control 75, 6 (1977); S. Clemmensen et al., Arch. Toxicol. 56, 43 (1984).
Reviews of use as fungicide in fish culture: N. C. Nelson, U.S. NTIS Report (PB-235450, 1974) 79 pp, C.A. 82, 150006p (1975), (PB-235451,
1974) 33 pp, C.A. 83, 1547j (1975); and as biological stain: Conn's Biological Stains, R. W. Horobin, J. A. Kiernan,
Eds. (BIOS Scientific Publishers Ltd, Oxford, UK, 10th ed., 2002)
189-190. Review of toxicological effects: S. Srivastava et
al., Aquat. Toxicol. 66, 310-329 (2004).
Properties
Green crystals with metallic luster. Very sol
in water; sol in alcohol, methanol, amyl alcohol. Water solns are
blue-green, absorption max 616.9 nm; yellow below pH 2. pK 6.90. LD50 in mice (mg/kg): 80 orally; 4.2 i.p. (Brock, Erhardt). LC50 (6 hr, 12°C) in bluegill sunfish,
channel catfish, rainbow trout, coho salmon (mg/l): 2.19 (pH 7.5), 0.960 (pH 7.5), 6.8 (pH 8.0), 3.0 (pH 7.5) (Bills, Marking).Derivative
Oxalate salt.
Nomenclature
CAS number: 2437-29-8
C
46H
50N
4.2HC
2O
4.C
2H
2O
4; mol wt 927.00.
C 67.37%, H 5.87%, N 6.04%, O 20.71%.
Properties
Sol to 4% in water, 5% in ethanol. Insol in xylene.Derivative
Leucomalachite green.
Nomenclature
CAS number: 129-73-7
Bis(
p-dimethylaminophenyl)phenylmethane.
C
23H
26N
2; mol wt 330.47.
C 83.59%, H 7.93%, N 8.48%.
Properties
Colorless, reduced form of malachite green. Practically
insol in water. Slightly sol in ethanol.Note
The term malachite green applies to
the oxalate as well as the chloride.Use
For directly dyeing silk, wool, jute and leather;
dyeing cotton after mordanting. Biological stain. Clinical reagent
(inorganic phosphate assay). As spot test reagent for detecting sulfurous
acid and cerium. As acid-base indicator: pH 0.0 yellow, 2.0 green;
11.6 green, 14 colorless.
Therapeutic Category (Veterinary)
Fungicide and parasiticide in fish.
