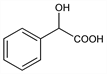
5717. Mandelic Acid
Nomenclature
CAS number: 90-64-2
α-Hydroxybenzeneacetic acid; dl-mandelic acid; racemic mandelic acid; α-hydroxy-α-toluic acid; α-hydroxyphenylacetic acid; phenylhydroxyacetic acid; phenylglycolic acid; amygdalic acid; amygdalinic acid; paramandelic acid; Uromaline. C8H8O3; mol wt 152.15.
C 63.15%, H 5.30%, O 31.55%.
Description and references
Prepd by the action of warm, dil alkali upon dichloroacetophenone: Org. Synth. 23, 48 (1943); by hydrolysis of mandelonitrile (prepd from benzaldehyde and hydrogen cyanide or from benzaldehyde, sodium bisulfite, and sodium cyanide): Org. Synth. coll. vol. I, 336 (1941); ibid. coll. vol. III, 538 (1955); L. F. Fieser, Organic Experiments (Boston, 1964) p 109. May be prepd by boiling amygdalin with HCl.
Properties
Orthorhombic plates from water, mp 119°. Darkens and dec on prolonged exposure to light. d 1.30. Can be distilled rapidly in vacuo at 2 mm without much decompn. Acid to litmus. pK (25°) 3.37. One gram dissolves in 6.3 ml water, 1 ml alc; freely sol in ether, isopropyl alcohol.Derivative
Calcium salt.Nomenclature
CAS number: 134-95-2
Calcium mandelate; Camdelate. C16H14CaO6; mol wt 342.36.
C 56.13%, H 4.12%, Ca 11.71%, O 28.04%.
Properties
Contains 88.88% mandelic acid. White powder. One gram dissolves in 80 ml boiling water. Slightly sol in cold water. Insol in alc. The aq soln is slightly acid.Derivative
Sodium salt.Nomenclature
CAS number: 114-21-6
Sodium mandelate. C8H7NaO3; mol wt 174.13.
C 55.18%, H 4.05%, Na 13.20%, O 27.56%.
Properties
Cryst powder; slight aromatic odor. Very sol in water; sol in alcohol; the aq soln is neutral or slightly alkaline to litmus.Therapeutic Category
Antiseptic (urinary).