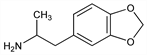
5765. MDA
Nomenclature
Description and references
Entactogen; synthetic amphetamine derivative with stimulant and hallucinogenic properties. Metabolite of MDMA, q.v. Prepn: C. Mannich, W. Jacobsohn, Ber. 43, 189 (1910); U. Braun et al., J. Pharm. Sci. 69, 192 (1980). Pharmacology: L. E. Markert, D. C. S. Roberts, Pharmacol. Biochem. Behav. 39, 569 (1991); K. M. Hegadoren et al., Psychopharmacology 118, 295 (1995). HPLC determn in whole blood: R. E. Michel et al., J. Neurosci. Methods 50, 61 (1993). GC/MS determn in hair: P. Kintz et al., J. Chromatogr. B 670, 162 (1995). Acute toxicity studies: P. N. Thiessen, D. A. Cook, Clin. Toxicol. 6, 193 (1973); H. F. Hardman et al., Toxicol. Appl. Pharmacol. 25, 299 (1973). Review of toxicity, mechanism of action, and psychoactive effects: R. P. Climko et al., Int. J. Psychiatry Med. 16, 359-372 (1986-1987).