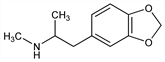
5767. MDMA
Nomenclature
Description and references
Entactogen; N-methyl derivative of MDA, q.v. (S)-Form possesses more potent CNS activity. Prepn: DE 274350 (1914 to E. Merck), C.A. 8, 3350 (1914); U. Braun et al., J. Pharm. Sci. 69, 192 (1980). Spectral and chromatographic analyses: K. Bailey et al., J. Assoc. Off. Anal. Chem. 58, 62 (1975). GC/MS determn in urine: B. K. Gan et al., J. Forensic Sci. 36, 1331 (1991). HPLC determn in whole blood: R. E. Michel et al., J. Neurosci. Methods 50, 61 (1993). Acute toxicity study: H. F. Hardman et al., Toxicol. Appl. Pharmacol. 25, 299 (1973). Series of articles on history, pharmacology, and neurotoxicity: Ann. N.Y. Acad. Sci. 600, 601-715 (1990). Review of biochemical actions: M. Rattray, Essays Biochem. 26, 77-87 (1991); of pharmacology, toxicology, and human experience: T. D. Steele et al., Addiction 89, 539-551 (1994); A. R. Green et al., Psychopharmacology 119, 247-260 (1995); H. Kalant, Can. Med. Assoc. J. 165, 917-928 (2001).