5775. Mechlorethamine
Nomenclature
Description and references
Nitrogen mustard alkylating agent; component of the MOPP chemotherapy protocol. Originally developed as a chemical warfare blister agent. Prepd by action of thionyl chloride on 2,2′-(methylimino)diethanol in trichloroethylene: Prelog, Stepan, Collect. Czech. Chem. Commun. 7, 93 (1935); Hanby, Rydon, J. Chem. Soc. 1947, 513; Abrams et al., J. Soc. Chem. Ind. London 68, 280 (1949); T. W. Doyle, D. M. Vyas in Kirk-Othmer Encyclopedia of Chemical Technology vol. 5 (John Wiley & Sons, New York, 4th ed., 1993) pp 876-879, 900-901. HPLC determn in pharmaceutical preparations: J. C. Reepmeyer, J. Chromatogr. A 1085, 262 (2005). Chemical weapons screening method and degradation profile in water: H.-C. Chua et al., ibid. 1102, 214 (2006). Environmental fate simulation: S. L. Bartelt-Hunt et al., Environ. Sci. Technol. 40, 4219 (2006). Effects of mustard alkylation on protein synthesis: A. Masta et al., Nucleic Acids Res. 23, 3508 (1995). Review of clinical experience in mycosis fungoides: E. C. Vonderheid, Int. J. Dermatol. 23, 180-186 (1984); Y. H. Kim et al., Arch. Dermatol. 139, 165-173 (2003); in Hodgkin's disease: C. Fermé et al., N. Engl. J. Med. 357, 1916-1927 (2007).
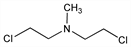
Properties
Mobile liquid. Faint odor of herring. Vesicant, necrotizing irritant. Never use without appropriate gas mask. Volatility at 25° = 3.581 mg/l. d425 1.118. mp -60°. bp18 87°; bp10 75°; bp5 64°; bp2 59°. Log P (octanol/water): 0.9. pKa: 6.8. Vapor pressure (25°): 0.427 mm Hg. Very slightly sol in water. Miscible with DMF, CS2, CCl4, many other organic solvents and oils. The undiluted liq dec on standing and forms polymeric quaternary ammonium salts which are insol in the free base.Derivative
Hydrochloride.Nomenclature
Properties
Light yellow brown, crystalline, hygroscopic leaflets from acetone or chloroform, mp 109-111°. Very sol in water; sol in alcohol. Initial pH of a 2% aq soln: 3.0-4.0. Dry crystals are stable at temps up to 40°. LD50 in rats (mg/kg): 1.1 i.v.; 1.9 s.c. See: W. P. Anslow et al., J. Pharmacol. Exp. Ther. 91, 224 (1947).Caution
Direct contact may cause severe skin and eye irritation; injury to deeper ocular structures, particularly the iris and lens. See Patty's Industrial Hygiene and Toxicology vol. 2E, G. D. Clayton, F. E. Clayton, Eds. (John Wiley & Sons, Inc., New York, 4th ed., 1994) pp 3297-3299. Mechlorethamine hydrochloride is reasonably anticipated to be a human carcinogen: Report on Carcinogens, Eleventh Edition (PB2005-104914, 2004) p III-192.Derivative
N-Oxide hydrochloride see Mechlorethamine Oxide Hydrochloride.Use
Base as a chemical warfare agent.Therapeutic Category
Hydrochloride as an antineoplastic.
Therapeutic Category (Veterinary)
Hydrochloride has been used as an antineoplastic.Keywords
Antineoplastic; Alkylating Agents; Nitrogen Mustards