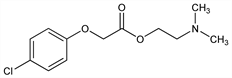
5780. Meclofenoxate
Nomenclature
CAS number: 51-68-3
(4-Chlorophenoxy)acetic acid 2-(dimethylamino)ethyl ester; dimethylaminoethyl p-chlorophenoxyacetate; centrophenoxine; meclofenoxane; acephen; ANP-235; Analux (Polfa); Cetrexin (Leciva); Proseryl (Funai). C12H16ClNO3; mol wt 257.71.
C 55.93%, H 6.26%, Cl 13.76%, N 5.44%, O 18.62%.
Description and references
Prepn: Rumpf, Thuillier, FR M398 (1962 to Centre Nat'l. Recherche Sci.), C.A. 57, 16768e (1962); Thuillier et al., Compt. Rend. 249, 2081 (1959); Thuillier, Rumpf, Bull. Soc. Chim. Fr. 1960, 1786. Pharmacology: Petkov, C.A. 65, 20717g (1966); Liberman, Farmakol. Toksikol. 30, 409 (1967). Auxin activity: Conti, Boll. Chim. Farm. 107, 325 (1968).
Derivative
Hydrochloride.Nomenclature
CAS number: 3685-84-5
Cellative (Tobishi); Clocete (Toyama); Lucidril (Dainippon); Methoxynal (Sankyo); Proserout (Funai); Brenal (Tanabe Seiyaku); Marucotol (Maruko); Helfergin (Promonta). C12H16ClNO3.HCl; mol wt 294.17.
C 48.99%, H 5.82%, Cl 24.10%, N 4.76%, O 16.32%.
Properties
Crystals from isopropyl alcohol or acetone, mp 135-139°. Sol in cold water. Sparingly sol in cold isopropyl alcohol, acetone. Practically insol in benzene, ether, chloroform. A 5% soln in water has pH 6. LD50 in mice (mg/kg): 330 i.v.; 1750 orally; 845 i.p. (Rumpf, Thuillier).Use
Plant growth regulator.Therapeutic Category
Cerebral stimulant.