5779. Meclofenamic Acid
Nomenclature
CAS number: 644-62-2
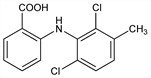
2-[(2,6-Dichloro-3-methylphenyl)amino]benzoic acid; N-(2,6-dichloro-m-tolyl)anthranilic acid; meclophenamic acid; CI-583; INF-4668; Arquel (Parke-Davis). C14H11Cl2NO2; mol wt 296.15.
C 56.78%, H 3.74%, Cl 23.94%, N 4.73%, O 10.80%.
Description and references
Prepn: Scherrer, Short, DE 1149015 C.A. 61, 1801d (1964) and US 3313848 (1963, 1967 to Parke, Davis); Juby et al., J. Med. Chem. 11, 111 (1968). Pharmacology and toxicology: Winder et al., J. Pharmacol. Exp. Ther. 148, 422 (1965); Winder, Ann. Phys. Med. 1966, 7; Kaump, ibid. 16. Series of articles on structure-activity relationships, mechanism of action, clinical studies: Arzneim.-Forsch. 33, 619-680 (1983). Effect on prostaglandin E receptor binding: M. C. P. Rees et al., Lancet 2, 541 (1988).
Properties
White crystals from acetone-water, mp 257-259°; also reported as mp 248-250°. Soly (mg/ml): water 0.03; 0.1N NaOH 28. pH of satd aq soln: ≈6.9.Derivative
Sodium salt monohydrate.Nomenclature
Lenidolor (Menarini); Meclodol (Parke-Davis); Meclomen (Parke-Davis); Movens (Inverni).C14H10Cl2NNaO2.H2O; mol wt 336.15.
C 50.02%, H 3.60%, Cl 21.09%, N 4.17%, Na 6.84%, O 14.28%.
Properties
mp 289-291°. Soly in water: 15 mg/ml (slightly turbid). pH 8.7.Therapeutic Category
Anti-inflammatory; antipyretic.
Therapeutic Category (Veterinary)
Anti-inflammatory.Keywords
Anti-inflammatory (Nonsteroidal); Aminoarylcarboxylic Acid Derivatives; Antipyretic