5844. Mepact
Nomenclature
Description and references
Synthetic, lipophilic analog of muramyl dipeptide, q.v. Immunostimulant promoting cancer cell destruction via in vivo macrophage activation. Prepn: L. Tarcsay et al., EP 25495; eidem, US 4406890 (1981, 1983 both to Ciba-Geigy). Synthesis of labeled form: D. E. Brundish, R. Wade, J. Labelled Compd. Radiopharm. 22, 29 (1985). Liposomal encapsulation: R. Natar et al., Methods Enzymol. 132, 594 (1986). Activation of tumoricidal properties in monocytes: E. S. Kleinerman et al., Cancer Res. 43, 2010 (1983). Potentiation of antitumor immunity: I. J. Fidler et al., J. Immunol. 138, 4509 (1987). Immunoassay in plasma: B. Gay et al., J. Biolumin. Chemilumin. 6, 73 (1991). Comparative pharmacokinetics of free and liposomal forms: B. Gay et al., J. Pharm. Sci. 82, 997 (1993). Mechanism of action study: T. Asano et al., J. Pharmacol. Exp. Ther. 268, 1032 (1994). Clinical study as chemotherapy adjuvant: P. A. Meyers et al., J. Clin. Oncol. 23, 2004 (2005). Review of phamacology and clinical studies as immune adjuvant in osteosarcoma: E. S. Kleinerman, Hematol. Oncol. Clin. North Am. 9, 927-938 (1995); A. Nardin et al., Curr. Cancer Drug Targets 6 123-133 (2006).
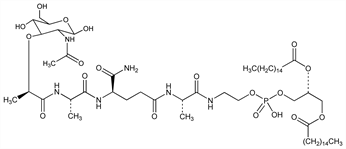
Derivative
Sodium salt.Nomenclature
Therapeutic Category
Antineoplastic (immunomodulator).
Keywords
Antineoplastic; Immunomodulators