5850. Mephenesin
Nomenclature
CAS number: 59-47-2
3-(2-Methylphenoxy)-1,2-propanediol; 3-(o-tolyloxy)-1,2-propanediol; 1,2-dihydroxy-3-(2-methylphenoxy)propane; α-(o-tolyl)glyceryl ether; glyceryl o-tolyl ether; o-cresyl glycerol ether; cresoxypropanediol; cresoxydiol; α:β-dihydroxy-γ-(2-methylphenoxy)propane; BDH-312; Atensin; Avosyl; Avoxyl; Curythan; Daserol; Decontractyl; Dioloxol (Reed & Carnrick); Glyotol; Glykresin; Lissephen (Abbott); Memphenesin; Mepherol; Mephesin; Mephson; Mervaldin (Lannett); Myanesin (BDH); Myanol; Myodetensine; Myolysin; Myopan; Myoserol; Myoten (Central Pharm.); Oranixon (Organon); Prolax; Relaxar; Relaxil; Renarcol; Rhex (Hobein); Sansdolor; Sinan; Spasmolyn; Stilalgin; Thoxidil; Tolansin; Tolcil; Tolhart; Tolosate; Toloxyn; Tolserol (BMS); Tolulexin; Tolulox; Tolyspaz (Conal); Walconesin. C10H14O3; mol wt 182.22.
C 65.91%, H 7.74%, O 26.34%.
Description and references
Prepn: P. Morch, Arch. Pharm. Chemi 54, 327 (1947), C.A. 42, 2058 (1948); GB 589821 (1947 to Carroll and Boake Roberts); of the carbamate: W. A. Lott, E. Pribyl, US 2609386 (1952 to Squibb). Pharmacology: F. M. Berger, W. Bradley, Br. J. Pharmacol. 1, 265 (1946); and toxicity data: P. E. Dresel, I. H. Slater, Proc. Soc. Exp. Biol. Med. 79, 286 (1952); A. P. Roszkowski, J. Pharmacol. Exp. Ther. 129, 75 (1960).
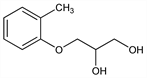
Properties
Crystals. Bitter taste. Produces numbness of the tongue. mp 70-71°. uv max (0.005% aq soln): 270 nm (E 0.395). Freely sol in alcohol, propylene glycol, chloroform. At 20° one part dissolves in 85 parts water, in 11 parts ether. Urea and its derivatives, particularly urethan, increase the water soly. One part dissolves in 60 parts of 5% urethan soln, in 40 parts of 10%, in 4.5 parts of 25%. Aq solns are stable, can be sterilized by heating, and are compatible and freely miscible with solns of sodium chloride, glucose, and derivatives of barbituric and thiobarbituric acids. pH of satd aq soln ≈6. LD50 in mice, rats, hamsters (mg/kg): 471, 283, 322 i.p.; 990, 945, 821 orally (Roszkowski). LD50 in mice (mM/kg): 2.83 i.p.; 10.53 orally (Dresel, Slater).Derivative
Carbamate.Nomenclature
CAS number: 533-06-2
Tolseram (BMS). C11H15NO4; mol wt 225.24.
C 58.66%, H 6.71%, N 6.22%, O 28.41%.
Properties
Crystals from water, mp about 93°. (Also a hemihydrate, mp 80-84°.) uv max (ethanol): 271, 277 nm (A1%1cm 72.7, 64.1). Has a lower water-solubility and a higher oil-solubility than mephenesin. Soly in water about 0.3%, in chloroform about 2.0%. Freely sol in alcohol. LD50 in mice (mM/kg): 2.77 i.p.; 7.67 orally (Dresel, Slater).Therapeutic Category
Muscle relaxant (skeletal).
Therapeutic Category (Veterinary)
Muscle relaxant.Keywords
Muscle Relaxant (Skeletal)