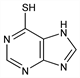
5871. 6-Mercaptopurine
Nomenclature
Description and references
Prepd from hypoxanthine and phosphorus pentasulfide: G. B. Elion et al., J. Am. Chem. Soc. 74, 411 (1952); G. H. Hitchings, G. B. Elion, US 2697709 (1954 to Burroughs Wellcome). Improved procedure: A. G. Beaman, R. K. Robins, J. Am. Chem. Soc. 83, 4042 (1961). Prepn from 7-aminothiazolo[5,4-d]pyrimidine: G. H. Hitchings, G. B. Elion, US 2721866 (1955 to Burroughs Wellcome). Metabolized in the body to 6-thiouric acid (6-mercapto-2,8-purinediol): G. B. Elion et al., J. Am. Chem. Soc. 81, 3042 (1959). Pharmacology: H. Froberg, M. S. Schencking, Arch. Toxicol. 32, 1 (1974). HPLC determn in body fluids: J. L. Rudy et al., Ann. Clin. Biochem. 25, 504 (1988). Clinical trial with methotrexate in acute lymphoblastic leukemia: S. Koizumi et al., Cancer 61, 1292 (1988). Comprehensive description: S. A. Benezra, P. R. B. Foss, Anal. Profiles Drug Subs. 7, 343-357 (1978). Review in treatment of Crohn's disease and ulcerative colitis: D. H. Present, Gastroenterol. Clin. North Am. 18, 57-71 (1989); in treatment of leukemia: B. A. Kamen, Semin. Hematol. 28, 12-14 (1991).
Derivative
Monohydrate.Properties
Yellow prisms from water. Becomes anhydrous at 140°, dec 313-314°. uv max (0.1N NaOH): 230, 312 nm (ε 14000, 19600); (0.1N HCl): 222, 327 nm (ε 9240, 21300); (methanol): 216, 329 nm (ε 8940, 19300). pKa1 7.77, pKa2 11.17. Insol in water, acetone, ether. Sol in hot ethanol, in alkaline solns with slow decompn. LD50 in mice, hamsters (mg/kg): 157, 364 i.p. (Froberg, Schencking).Therapeutic Category
Antineoplastic; immunosuppressant.
Therapeutic Category (Veterinary)
Antineoplastic.Keywords
Antineoplastic; Antimetabolites; Purine Analogs; Immunosuppressant