5904. Mesalamine
Nomenclature
Description and references
Prepn by reduction of m-nitrobenzoic acid with Zn dust and HCl: H. Weil et al., Ber. 55B, 2664 (1922); by electrolytic reduction: Le Guyader, Peltier, Compt. Rend. 253, 2544 (1961). Use in manufacture of dyes: GB 751386 (1956 to J. R. Geigy). Identification as active metabolite of sulfasalazine, q.v.: A. K. Azad Khan et al., Lancet 2, 892 (1977); P. A. M. Van Hees et al., Gut 21, 632 (1980). HPLC determn in serum: E. Brendel et al., J. Chromatogr. 385, 299 (1987). Bioavailability, plasma level and excretion: S. N. Rasmussen et al., Gastroenterology 83, 1062 (1982). Clinical evaluation in Crohn's disease: eidem, ibid. 85, 1350 (1983). Clinical trials in ulcerative colitis: M. Campieri et al., Lancet 2, 270 (1981); L. S. Friedman et al., Am. J. Gastroenterol. 81, 412 (1986); in comparison with olsalazine, q.v.: S. S. Rao et al., Scand. J. Gastroenterol. 22, 332 (1987). Brief review: G. Friedman, Am. J. Gastroenterol. 81, 141-144 (1986). Review of clinical efficacy in colonic diverticulosis: A. Tursi, Expert Opin. Pharmacother. 6, 69-74 (2005).
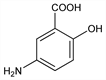
Properties
White to pinkish crystals, dec ≈280°. Slightly sol in cold water, alc; more sol in hot water; sol in HCl.Use
In manuf of light-sensitive paper, azo and sulfur dyes.Therapeutic Category
Anti-inflammatory (gastrointestinal).
Keywords
Anti-inflammatory (Gastrointestinal); Anti-inflammatory (Nonsteroidal); Salicylic Acid Derivatives