5903. Mesaconic Acid
Nomenclature
CAS number: 498-24-8
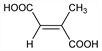
(E)-2-Methyl-2-butenedioic acid; methylfumaric acid; trans-1-propene-1,2-dicarboxylic acid. C5H6O4; mol wt 130.10.
C 46.16%, H 4.65%, O 49.19%.
Description and references
Prepd by heating citraconic anhydride with nitric acid: Shriner et al., Org. Synth. 11, 74 (1931); from ethyl itaconate: Jones et al., J. Chem. Soc. 1954, 1865; from N,N-diethyl-5-diethylamino-2,3-dihydro-3-furamide: Sauer et al., J. Am. Chem. Soc. 81, 693 (1959). Obtained almost instantly by the action of sunlight on citraconic acid in ether + chloroform in the presence of a little bromine: Fittig, Langworthy, Ber. 26, 46 (1893); Ann. 304, 119, 149 (1899); by action of β-radiation on citraconic acid + bromine: Lavigne, Levine, US 2979445 (1961 to California Res. Corp.).
Properties
Orthorhombic needles from alcohol, monoclinic tablets from ethyl acetate, mp 204-205°. d 1.466. Sublimes. bp 250° (decompn). K1 at 25° = 8.5 × 104; K2 = 15 × 106. 100 g water dissolves 2.7 g at 18° and 117.9 g at the boiling point; 100 g 90% alc dissolves 30.6 g at 17° and 95.7 g at the boiling point. Sol in ether; sparingly sol in chloroform, carbon disulfide and ligroin.Derivative
Dimethyl ester.C7H10O4; mol wt 158.15.
C 53.16%, H 6.37%, O 40.47%.
Properties
Liq, bp 206°. d421 vac 1.12011. nD20 1.45575. Sol in 122 parts water at 15°.Derivative
Diethyl ester.C9H14O4; mol wt 186.21.
C 58.05%, H 7.58%, O 34.37%.