5943. Methacycline
Nomenclature
CAS number: 914-00-1
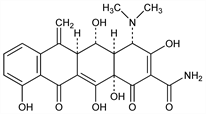
[4S-(4α,4aα,5α,5aα,12aα)]-4-Dimethylamino-1,4,4a,5,5a,6,11,12a-octahydro-3,5,10,12,12a-pentahydroxy-6-methylene-1,11-dioxo-2-naphthacenecarboxamide; 6-methyleneoxytetracycline; 6-methylene-5-hydroxytetracycline; metacycline. C22H22N2O8; mol wt 442.42.
C 59.73%, H 5.01%, N 6.33%, O 28.93%.
Description and references
Broad spectrum, semi-synthetic antibiotic related to tetracycline, q.v. Prepn from oxytetracycline, q.v.: R. K. Blackwood et al., US 2984686 (1961 to Pfizer); eidem, J. Am. Chem. Soc. 83, 2773 (1961); 85, 3943 (1963). Solubility data: J. R. Marsh, P. J. Weiss, J. Assoc. Off. Anal. Chem. 50, 257 (1967). Toxicity data: E. I. Goldenthal, Toxicol. Appl. Pharmacol. 18, 185 (1971). Comparative clinical study with ampicillin, q.v., in chronic bronchitis: S. Chodosh et al., Chest 69, 587 (1976).
Derivative
Hydrochloride.Nomenclature
CAS number: 3963-95-9
Adriamicina (Farmochim. Ital.); Ciclobiotic (Beta); Germiciclin (Mendelejeff); Metadomus (Med. Domus); Metilenbiotic (Coli); Londomycin; Optimycin (Novartis); Physiomycine; Rindex (Sidus); Rondomycin (Wallace). C22H22N2O8.HCl; mol wt 478.88.
C 55.18%, H 4.84%, N 5.85%, O 26.73%, Cl 7.40%.
Properties
Obtained as crystals containing 0.5 mol water and 0.5 mol methanol from methanol + acetone + concd HCl + ether. Yellow, crystalline powder, dec ≈205°. Bitter taste. Soluble in water, sparingly sol in alcohol. Practically insol in ether, chloroform. uv max (methanol + 0.01N HCl): 253, 345 nm (log ε 4.37, 4.19). LD50 in rats, mice (mg/kg): 252, 288 i.p. (Goldenthal).Therapeutic Category
Antibacterial.
Keywords
Antibacterial (Antibiotics); Tetracyclines