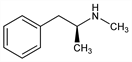
5948. Methamphetamine
Nomenclature
Description and references
Central stimulant. Can be prepd by reducing ephedrine or pseudoephedrine: Emde, Helv. Chim. Acta 12, 365 (1929). Prepn by reducing the condensation product of benzyl methyl ketone and methylamine: A. Ogata, J. Pharm. Soc. Jpn. 451, 751 (1919), C.A. 14, 745 (1920). Synthesis from d-phenylalanine: D. B. Repke et al., J. Pharm. Sci. 67, 1167 (1978). Stereochemistry-pharmacology aspects: Patil et al., J. Pharmacol. Exp. Ther. 155, 1, 13 (1967). Toxicity data: A. M. Lands et al., J. Pharmacol. Exp. Ther. 89, 382 (1947). Review of clinical trials in bulimia: H. G. Pope, Jr., J. I. Hudson, J. Clin. Psychiatry 47, 339 (1986). Review of pharmacology and abuse potential: A. K. Cho, Science 249, 631-634 (1990).
Derivative
Hydrochloride.Nomenclature
Properties
Crystals, mp 170-175°. Bitter taste. [α]D25 +14 to +20°. Sol in water, alcohol, chloroform. Practically insol in ether. A 1% aq soln is neutral or slightly acid to litmus. LD50 i.p. in mice: 70 mg/kg (Lands).Note
This is a controlled substance (stimulant): 21 CFR, 1308.12.Therapeutic Category
Anorexic. In attention deficit disorder with hyperactivity.
Therapeutic Category (Veterinary)
Sympathomimetic, CNS stimulant.Keywords
Anorexic; CNS Stimulant