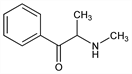
5963. Methcathinone
Nomenclature
Description and references
Oxidation product of ephedrine possessing amphetamine-like central stimulant properties. Prepn: J. F. Hyde et al., J. Am. Chem. Soc. 50, 2287 (1928); K. Y. Zhingel et al., J. Forensic Sci. 36, 915 (1991). Prepn of enantiomers and chromatographic analysis: J. DeRuiter et al., J. Chromatogr. Sci. 32, 552 (1994). Comparative pharmacology: R. Young, R. A. Glennon, Pharmacol. Biochem. Behav. 45, 229 (1993). Pharmacology of enantiomers: R. A. Glennon et al., ibid. 50, 601 (1995). Absorption and metabolism: S. L. Markantonis et al., J. Pharm. Pharmacol. 38, 515 (1986). GLC/MS determn in urine: eidem, Chim. Chron. 14, 211 (1985). Impact on public health: T. S. Emerson, J. E. Cisek, Ann. Emerg. Med. 22, 1897 (1993).
Derivative
Hydrochloride.Nomenclature
Properties
Crystals from alcohol-acetone, mp 176-177°.Derivative
l-Form hydrochloride.Description and references
Prepn: Y. J. L'Italien, M. C. Rebstock, US 2802865 (1957 to Parke, Davis).