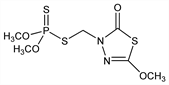
5970. Methidathion
Nomenclature
CAS number: 950-37-8
Phosphorodithioic acid S-[(5-methoxy-2-oxo-1,3,4-thiadiazol-3(2H)-yl)methyl] O,O-dimethyl ester; phosphorodithioic acid O,O-dimethyl ester S-ester with 4-(mercaptomethyl)-2-methoxy-Δ2-1,3,4-thiadiazolin-5-one; dithiophosphoric acid O,O′-dimethyl-S-[(5-methoxy-1,3,4-thiadiazol-2(3H)-one-3-yl)methyl] ester; O,O′-dimethyl-S-[(2-methoxy-1,3,4-thiadiazol-5(4H)-one-4-yl)methyl] dithiophosphate; GS-13005; Supracide (Syngenta). C6H11N2O4PS3; mol wt 302.33.
C 23.84%, H 3.67%, N 9.27%, O 21.17%, P 10.25%, S 31.82%.
Description and references
Organophosphate insecticide; cholinesterase inhibitor. Synthesis and degradation products: Rüfenacht, Helv. Chim. Acta 51, 518 (1968) DOI PubMed; FR 1335755 (1963 to Geigy), C.A. 60, 1764g (1964). Metabolism in rats: Esser et al., Helv. Chim. Acta 51, 513 (1968) DOI PubMed. Acute toxicity: T. B. Gaines, R. E. Linder, Fundam. Appl. Toxicol. 7, 299 (1986) DOI PubMed. Voltammetric determn in water samples: G. Erdogdu, J. Anal. Chem. 61, 673 (2006) DOI. Photodegradation studies in soil: L. Sánchez et al., Chemosphere 59, 969 (2005) DOI. Carcinogenicity studies: J. A. Quest et al., Regul. Toxicol. Pharmacol. 12, 117 (1990) DOI PubMed.