5981. Methohexital
Nomenclature
CAS number: 151-83-7
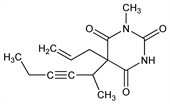
1-Methyl-5-(1-methyl-2-pentyn-1-yl)-5-(2-propen-1-yl)-2,4,6(1H,3H,5H)-pyrimidinetrione; α-dl-5-allyl-1-methyl-5-(1-methyl-2-pentynyl)barbituric
acid; α-dl-1-methyl-5-(1-methyl-2-pentynyl)-5-allylbarbituric
acid; methohexitone. C14H18N2O3; mol wt 262.30.
C 64.11%, H 6.92%, N 10.68%, O 18.30%.
Description and references
Barbituric acid hypnotic. Prepn of the α-racemate: W. J. Doran, US 2872448 (1959 to Lilly). Resolution of enantiomers: idem, J. Org. Chem. 25, 1737 (1960). Enantioselective synthesis and activity of isomers: H. Brunner, Chirality 13, 420 (2001); idem et al., Eur. J. Org. Chem. 2003, 855. HPLC determn in plasma: M. J. Avram, T. C. Krejcie, J. Chromatogr. 414, 484 (1986). Clinical pharmacokinetics: Y. Le Normand et al., Br. J. Clin. Pharmacol. 26, 589 (1988). Review of use in dental anesthesia: C. H. Martone et al., Anesth. Prog. 38, 195-199 (1991); in orthopedic procedural sedation: T. Austin et al., J. Emerg. Med. 24, 315-318 (2002).
Properties
Crystals from dil ethanol as the α-dl-racemate, mp 96°. Very slightly sol in water; slightly sol in ethanol, chloroform, dil alkalies.Derivative
Sodium salt.Nomenclature
CAS number: 309-36-4
Brevital (Monarch); Brevimytal (Lilly); Brietal (Lilly). C14H17N2NaO3; mol wt 284.29.
C 59.15%, H 6.03%, N 9.85%, Na 8.09%, O 16.88%.
Properties
White crystalline powder. Freely soluble in water.Note
This is a controlled substance (depressant): 21 CFR, 1308.14.Therapeutic Category
Anesthetic (intravenous).
Therapeutic Category (Veterinary)
Anesthetic (intravenous).Keywords
Anesthetic (Intravenous)