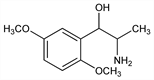
5987. Methoxamine
Nomenclature
CAS number: 390-28-3
α-(1-Aminoethyl)-2,5-dimethoxybenzenemethanol; α-(1-aminoethyl)-2,5-dimethoxybenzyl alcohol; 2-amino-1-(2,5-dimethoxyphenyl)-1-propanol; β-hydroxy-β-(2,5-dimethoxyphenyl)isopropylamine; β-(2,5-dimethoxyphenyl)-β-hydroxyisopropylamine; 2,5-dimethoxynorephedrine. C11H17NO3; mol wt 211.26.
C 62.54%, H 8.11%, N 6.63%, O 22.72%.
Description and references
α1-Adrenergic agonist. Prepn: Baltzly et al., US 2359707 (1944 to Burroughs Wellcome). Metabolism: A. Klutch, M. Bordun, J. Med. Chem. 10, 860 (1967). Clinical pharmacology: N. T. Smith, C. Whitcher, Anesthesiology 28, 735 (1967); P. D. Snashall et al., Clin. Sci. Mol. Med. 54, 283 (1978). HPLC determn in plasma: I. A. Al-Meshal et al., J. Liq. Chromatogr. 12, 1589 (1989). Therapeutic use: P. M. C. Wright et al., Anesth. Analg. 75, 56 (1992); L. Cabanes et al., N. Engl. J. Med. 326, 1661 (1992). Comprehensive description: A. M. Al-Obaid, M. M. El-Domiaty, Anal. Profiles Drug Subs. 20, 399-431 (1991).
Derivative
Hydrochloride.Nomenclature
CAS number: 61-16-5
Vasoxine (Burroughs Wellcome); Vasoxyl (Burroughs Wellcome); Vasylox (Burroughs Wellcome). C11H17NO3.HCl; mol wt 247.72.
C 53.33%, H 7.32%, N 5.65%, O 19.38%, Cl 14.31%.
Properties
Crystals, mp 212-216°. pKa (25°C) 9.2. Very sol in water: One gram dissolves in 2.5 ml water, in 12 ml ethanol. Practically insol in ether, benzene, chloroform. pH of a 2% aq soln between 4.5 and 5.5.Therapeutic Category
Antihypotensive.
Keywords
α-Adrenergic Agonist; Antihypotensive