5988. Methoxsalen
Nomenclature
Description and references
Naturally occurring analog of psoralen, q.v., found in spp. of Leguminosae, Umbelliferae, and Rutaceae. Isolation: Priess, Ber. Dtsch. Pharm. Ges. 21, 227 (1911); Thoms, Ber. 44, 3325 (1911); 45, 3705 (1912); Jois et al., J. Indian Chem. Soc. 10, 41 (1933); Spth et al., Ber. 73, 1361 (1933); Schonberg, Sina, Nature 160, 468 (1947); 161, 481 (1948); J. Am. Chem. Soc. 72, 4826 (1950). Synthesis: Spth, Pailer, Ber. 69, 767 (1936); Lagercrantz, Acta Chem. Scand. 10, 647 (1956); Stanley, Vannier, US 2889337 (1959 to USDA); P. Nore, E. Honkanen, J. Heterocycl. Chem. 17, 985 (1980). Use in treatment of psoriasis and mycosis fungoides: J. A. Parrish et al., Int. J. Dermatol. 19, 379 (1980). Acute toxicity data: Hakim et al., J. Pharmacol. Exp. Ther. 131, 394 (1961). Phototoxicity study: A. Kornhauser et al., Science 217, 733 (1982). Comprehensive description: M. A. Loutfy, M. A. Hassan, Anal. Profiles Drug Subs. 9, 427-454 (1980). Review of use in photochemotherapy: T. F. Anderson, J. J. Voorhees, Annu. Rev. Pharmacol. Toxicol. 20, 235-258 (1980); Acta Derm. Venereol. Suppl. 106, 9-42 (1982).
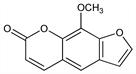
Properties
Silky needles from hot water or benzene + petr ether, long rhombic prisms from alcohol + ether. mp 148°. Odorless. Bitter taste followed by tingling sensation. pH 5.5. uv max: 219, 249, 300 nm (log ε 4.32, 4.35, 4.06). Practically insol in cold water. Sparingly sol in boiling water, liquid petrolatum, ether. Sol in boiling alcohol, acetone, acetic acid, vegetable fixed oils, propylene glycol, benzene. Freely sol in chloroform. Sol in aq alkalies with ring cleavage, but is reconstituted upon neutralization. LD50 i.p. in rats: 470 ±30 mg/kg (Hakim).Caution
Methoxsalen with ultraviolet A (long wave light) therapy (PUVA) is listed as a known human carcinogen: Report on Carcinogens, Eleventh Edition (PB2005-104914, 2004) p III-165.Therapeutic Category
Pigmentation agent.
Keywords
Pigmentation Agent