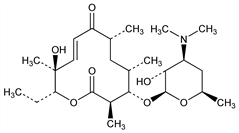
6135. Methymycin
Nomenclature
CAS number: 497-72-3
C25H43NO7; mol wt 469.61.
C 63.94%, H 9.23%, N 2.98%, O 23.85%.
Description and references
Antibiotic substance produced by a streptomycete from soil near Oswego, N.Y. Has a macrolide structure (12-membered lactone ring, compare Picromycin). Isoln and antibacterial activity: Donin et al., Antibiot. Annu. 1, 179 (1953-4). Production using Streptomyces venezuelae cultures: Dutcher et al., US 2916483 (1959 to Olin Mathieson). Structure: C. Djerassi, J. A. Zderic, J. Am. Chem. Soc. 78, 6390 (1956). Absolute configuration: Rickards, Smith, Tetrahedron Lett. 1970, 1025; Manwaring et al., ibid. 1029. Biosynthesis: Birch et al., J. Chem. Soc. 1964, 5274. Synthesis: Masamune et al., J. Am. Chem. Soc. 97, 3512 (1975). Possible mechanism of action: Wilhelm et al., Antimicrob. Agents Chemother. 1967, 236.
Properties
Prisms from abs ethanol, mp 195.5-197°. [α]D22 +61° (c = 0.7 in methanol), +74° (c = 1.1 in chloroform). Basic substance, pKb′ 5.7. uv max (methanol): 223, 322 nm (ε 10500, 47). Very slightly sol in water, hexane. Sol in methanol, acetone, chloroform, dil acids. Moderately sol in ethanol, ether.Derivative
Sulfate.(C25H43NO7)2.H2SO4; mol wt 1037.30.
C 57.89%, H 8.55%, N 2.70%, O 27.76%, S 3.09%.
Properties
Crystals from methanol + acetone. Sol in water, methanol.Derivative
Acid sulfate.C25H43NO7.H2SO4; mol wt 567.69.
C 52.89%, H 7.99%, N 2.47%, O 31.00%, S 5.65%.