6178. Miconazole
Nomenclature
CAS number: 22916-47-8
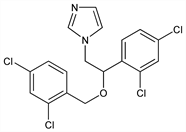
1-[2-(2,4-Dichlorophenyl)-2-[(2,4-dichlorophenyl)methoxy]ethyl]-1H-imidazole; 1-[2,4-dichloro-β-[(2,4-dichlorobenzyl)oxy]phenethyl]imidazole. C18H14Cl4N2O; mol wt 416.13.
C 51.95%, H 3.39%, Cl 34.08%, N 6.73%, O 3.84%.
Description and references
Prepn: E. F. Godefroi et al., J. Med. Chem. 12, 784 (1969); E. F. Godefroi, J. Heeres, DE 1940388; eidem, US 3717655 (1970, 1973 to Janssen). Clinical evaluation: Brugmans et al., Arch. Dermatol. 102, 428 (1970); Godts et al., Arzneim.-Forsch. 21, 256 (1971). Review: P. Janssen, W. Van Bever, in Pharmacological and Biochemical Properties of Drug Substances vol. 2, M. E. Goldberg, Ed. (Am. Pharm. Assoc., Washington, DC, 1979) pp 333-354; R. C. Heel et al., Drugs 19, 7-30 (1980).
Derivative
Nitrate.Nomenclature
CAS number: 22832-87-7
R-14889; Aflorix (Gramon); Albistat (Ortho); Andergin (ISOM); Brentan (Janssen); Conoderm (C-Vet); Conofite (Mallinckrodt); Daktar (Janssen); Daktarin (Janssen); Deralbine (Andromaco); Dermonistat (Ortho); Epi-Monistat (Cilag); Florid (Mochida); Fungiderm (Janssen); Fungisdin (Isdin); Gyno-Daktarin (Janssen); Gyno-Monistat (Cilag-Chemie); Micatin (J & J); Miconal Ecobi (Ecobi); Micotef (LPB); Monistat (Cilag-Chemie); Prilagin (Gambar); Vodol (Andromaco). C18H14Cl4N2O.HNO3; mol wt 479.14.
C 45.12%, H 3.16%, Cl 29.60%, N 8.77%, O 13.36%.
Properties
Crystals, mp 170.5° (Godefroi, Heeres, 1970); 184-185° (Godefroi).Derivative
(+)-Form nitrate.Properties
mp 135.3°. [α]D20 +59° (methanol).Derivative
()-Form nitrate.Properties
mp 135°. [α]D20 58° (methanol).Therapeutic Category
Antifungal (topical).
Therapeutic Category (Veterinary)
Antifungal (topical).Keywords
Antifungal (Synthetic); Imidazoles