6181. Micronomicin
Nomenclature
Description and references
Gentamicin C complex antibiotic produced by Micromonospora sagamiensis var. nonreducans: T. Nara et al., DE 2326781 (1973 to Kyowa), C.A. 80, 58389b (1974); eidem, US 4045298 (1977 to Abbott). Isoln, physicochemical and antibacterial properties: R. Okachi et al., J. Antibiot. 27, 793 (1974). Structure: R. S. Egan et al., ibid. 28, 29 (1975). Identity with gentamicin C2b: P. J. L. Daniels et al., ibid. 35. Pharmacology: T. Hashimoto et al., Jpn. J. Antibiot. 30, 362 (1977), C.A. 89, 17113z (1978). Toxicity studies: T. Hara et al., ibid. 386-449, C.A. 89, 209205c-8f (1978). Biosynthesis: H. Kase et al., J. Antibiot. 35, 1 (1982); eidem, Agric. Biol. Chem. 46, 515 (1982). Series of articles on pharmacology, metabolism and clinical studies: Chemotherapy (Tokyo) 25, 1801-2287 (1977). See also Gentamicin.
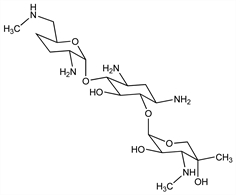
Properties
White amorphous powder, mp 260° (dec). [α]D20 +116° (c = 1 in water). Sol in water, methanol. Insol in chloroform, ethyl acetate, benzene, petr ether. LD50 i.v. in mice: 93 mg/kg (Okachi).Derivative
Sulfate.Nomenclature
6′-N-Methylgentamicin C1a hemipentasulfate; Sagamicin (Kyowa); Santemycin (Santen).Therapeutic Category
Antibacterial.
Keywords
Antibacterial (Antibiotics); Aminoglycosides