4392. Gentamicin
Nomenclature
Description and references
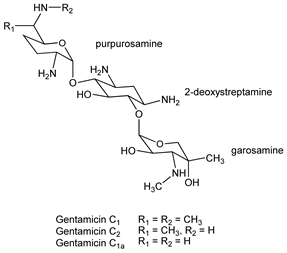
Antibiotic complex produced by fermentation of Micromonospora purpurea or M. echinospora and variants thereof: M. J. Weinstein et al., Antimicrob. Agents Chemother. 1963, 1. Isoln, purification and characterization: J. P. Rosselet et al., ibid. 14. Industrial pats.: G. M. Luedemann, M. J. Weinstein; Charney, US 3091572; US 3136704 (1963, 1964, both to Schering). Consists of three closely related components, gentamicins C1, C2, C1a, and also gentamicin A which differs from the other members of the complex but is similar to kanamycin C, q.v. Separation of gentamicin C components: H. Maehr, C. P. Schaffner, J. Chromatogr. 30, 572 (1967); Wagman et al., ibid. 34, 210 (1968). Structures contain 2-deoxystreptamine, q.v., linked to two saccharide units, these being garosamine and a purpurosamine in the C series gentamicins. Structure studies: D. J. Cooper et al., J. Chem. Soc. C 1971, 960, 2876, 3126. Structure of gentamicin A: H. Maehr, C. P. Schaffner, J. Am. Chem. Soc. 89, 6787 (1967); 92 1697 (1969). Sepn and structures of gentamicins A1 to A4: Nagabhushan et al., J. Org. Chem. 40, 2830, 2835 (1975). Synthetic studies: W. Meyer zu Reckendorf, Bischof, Ber. 105, 2546 (1972); M. Chmielewski et al., Carbohydr. Res. 70, 275 (1979). Review of activity, toxicity and clinical pharmacology: J. Black et al., Antimicrob. Agents Chemother. 1963, 138-147. Comprehensive description: B. E. Rosenkrantz et al., Anal. Profiles Drug Subs. 9, 295-340 (1980). Determn in serum by immunoassay: H. A. Holt et al., J. Antimicrob. Chemother. 34, 747 (1994). Review of clinical use: S. B. Shrimpton et al., ibid. 31, 599-606 (1993). Clinical effect on chloride transport in cystic fibrosis: M. Wilschanski et al., Am. J. Respir. Crit. Care Med. 161, 860 (2000).
Properties
White amorphous powder, mp 102-108°. [α]D25 +146°. Freely sol in water; sol in pyridine, DMF, in acidic media with salt formation; moderately sol in methanol, ethanol, acetone. Practically insol in benzene, halogenated hydrocarbons.Derivative
Gentamicin C1.Nomenclature
Properties
mp 94-100°. [α]D25 +158°.Derivative
Gentamicin C2.Nomenclature
Properties
mp 107-124°. [α]D25 +160°.Derivative
Gentamicin C1a.Nomenclature
Derivative
Gentamicin A.Nomenclature
Derivative
Hydrochloride.Properties
mp 194-209°. [α]D25 +113°. Freely sol in water, methanol; slightly in ether. Practically insol in other organic solvents.Derivative
C complex sulfate.Nomenclature
Properties
White, hygroscopic powder, mp 218-237°. [α]D25 +102°. Sol in ethylene glycol, formamide. LD50 in mice (mg base/kg): 430 i.p.; 485 s.c.; 75 i.v.; >9050 orally (Black).Therapeutic Category
Antibacterial.
Therapeutic Category (Veterinary)
Antibacterial.Keywords
Antibacterial (Antibiotics); Aminoglycosides