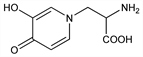
Nomenclature
CAS number: 500-44-7
α-Amino-3-hydroxy-4-oxo-1(4
H)-pyridinepropanoic
acid; 3-hydroxy-4-oxo-1(4
H)-pyridinealanine; β-[
N-(3-hydroxy-4-pyridone)]-α-aminopropionic acid; leucenol; leucenine; leucaenine; leucaenol.
C
8H
10N
2O
4; mol wt 198.18.
C 48.48%, H 5.09%, N 14.14%, O 32.29%.
Description and references
A naturally occurring amino acid found in large
quantities in the seeds and foliage of the legume genera Mimosa and Leucena. Isoln from Leucena glauca (Willd.)
Benth., Leguminosae: Renz, Z.
Physiol. Chem. 244, 153 (1936); Mascré, Compt. Rend. 204, 890 (1937);
improved large-scale isoln: N. K. Hart et al., Heterocycles 7, 265 (1977). Structure:
Adams, Jones, J. Am. Chem. Soc. 69, 1803 (1947). Synthesis: Adams, Johnson, ibid. 71, 705 (1949). Identity
of mimosine and leucenol: Kleipool, Wibaut, Rec. Trav. Chim. 69, 37 (1950); Wibaut, Schuhmacher, ibid. 71, 1017 (1952). Crystal
structure: A. Mostad et al., Acta
Chem. Scand. 27, 164 (1973). Shown to cause
inhibition of hair growth and loss of hair in mice: Crounse et al., Nature 194, 694 (1962). Explanation of the toxicity in animals: J. F. Thompson et al., Annu. Rev. Biochem. 38, 137 (1969).
Derivative
dl-Form.
Properties
Crystals from water, mp 235-236°. Slightly sol in water, much less
sol in methanol and ethanol. Practically insol in the higher alcohols,
in dioxane, ethyl acetate, ether, benzene, chloroform, glacial acetic
acid, pyridine, Cellosolve. Sol in dil acids or bases and may be
recovered from these solns by adjusting the pH so that it is just
acid to bromcresol green. uv max: 282 nm (log ε 4.23).Derivative
dl-Hemihydrate.
Properties
Crystals, darkens at 215-226°, mp 227-228°.Derivative
l-Form.
Properties
Crystals from water, mp 225°. [α]D22 -20°.Derivative
Hydrochloride.
C
8H
11ClN
2O
4; mol wt 234.64.
C 40.95%, H 4.73%, Cl 15.11%, N 11.94%, O 27.27%.
Properties
Dec 175°, sol in water.Derivative
Hydrobromide.
C
8H
11BrN
2O
4; mol wt 279.09.
C 34.43%, H 3.97%, Br 28.63%, N 10.04%, O 22.93%.
Properties
Dec 179.5°, sol in water.Use
Depilatory agent, Hegarty
et al., Aust. J. Agric. Res. 15, 153 (1964),
C.A. 60, 16405e (1964).
