Nomenclature
CAS number: 480-16-0
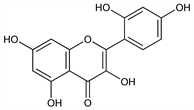
2-(2,4-Dihydroxyphenyl)-3,5,7-trihydroxy-4
H-1-benzopyran-4-one; 2′,3,4′,5,7-pentahydroxyflavone; 2′-hydroxypelargidenolon 1522; C.I. Natural Yellow 8; C.I. Natural Yellow 11; C.I. 75660.
C
15H
10O
7; mol wt 302.24.
C 59.61%, H 3.33%, O 37.06%.
Description and references
In wood of old fustic (Chlorophora tinctoria (L.) Gaud., Moraceae), also called Cuba wood, or yellow
Brazil wood. The wood of the Osage orange tree also contains morin
and maclurin. Isoln: Perkin, Pate, J. Chem.
Soc. 67, 649 (1895); Rolland, Teintex 3, 460 (1938). Synthesis
by condensing phloracetophenone dimethyl ether with 2,4-dimethoxybenzaldehyde:
von Kostanecki et al., Ber. 39, 625, 4014, 4022 (1906). Comprehensive list of refs
in Colour Index vol. 4 (3rd ed., 1971) p 4636.
Properties
Crystallizes with 1 or 2 mols water. Anhydr needles
from abs alcohol, dec 285-290°. One gram dissolves in 4 liters water
at 20°, in 1060 ml boiling water. Freely sol in alcohol; slightly
sol in ether, acetic acid. Sol in aq alkaline solns with intense
yellow color which turns brown on exposure to air. Absorpion spectrum:
Grinbaumowna, Marchlewski, Biochem. Z. 290, 261 (1937).Derivative
Calico yellow.
Properties
Obtained by the action of bisulfite on fustic
extract consists mainly of the bisulfite compound of morin, C15H10O7 + NaHSO3, and is used
in calico printing. The calico yellow of commerce is a good source
of morin for the laboratory. The bisulfite is removed with HCl.Use
As a spot test reagent for Al, Be, Zn, Ga, In, and
Sc salts: Feigl,
Spot Tests (New
York, 1954); Beck,
Mikrochim. Acta 2, 287 (1937). As luminescence indicator: Kocsis, Zádor,
Z. Anal. Chem. 124, 42-45 (1942).
As a textile dye, morin dyes wool mordanted with chromium: olive
yellow; mordanted with aluminum: yellow; mordanted with tin: lemon
yellow; mordanted with iron: deep olive brown.
