6268. Moricizine
Nomenclature
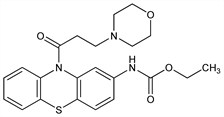
CAS number: 31883-05-3
[10-[3-(4-Morpholinyl)-1-oxopropyl]-10H-phenothiazin-2-yl]carbamic
acid ethyl ester; 10-(3-morpholinopropionyl)phenothiazine-2-carbamic acid ethyl
ester; ethmosine; ethmozin; moracizine; EN-313. C22H25N3O4S; mol wt 427.52.
C 61.81%, H 5.89%, N 9.83%, O 14.97%, S 7.50%.
Description and references
Sodium channel blocker. Prepn: A. Gritsenko et al., DE 2014201 (1971 to Acad. Med. Sci., USSR), C.A. 76, 34273a (1972); eidem, US 3740395 (1973). Pharmacology and toxicity: N. V. Kaverina et al., Russ. Pharmacol. Toxicol. 35, 74 (1972). Mechanism of action study: T. Yamane et al., Br. J. Pharmacol. 108, 812 (1993). HPLC determn in plasma: C. C. Whitney et al., J. Pharm. Sci. 70, 462 (1981). Symposium on pharmacology, pharmacokinetics, clinical safety and efficacy: Am. J. Cardiol. 65, 1D-71D (1990). Review: C. A. Clyne et al., N. Engl. J. Med. 327, 255 (1992).
Properties
Crystals, mp 156-157°. pKa 6.4.Derivative
Hydrochloride.Nomenclature
CAS number: 29560-58-5
Ethmozine (Roberts). C22H25N3O4S.HCl; mol wt 463.98.
C 56.95%, H 5.65%, N 9.06%, O 13.79%, S 6.91%, Cl 7.64%.
Properties
Crystals from dichloroethane, mp 189° (dec). Sol in water, alcohol. LD50 in mice, rats (mg/kg): 36, 12 i.v.; in mice (mg/kg): 131 i.p. (Kaverina).Therapeutic Category
Antiarrhythmic (class I).
Keywords
Antiarrhythmic