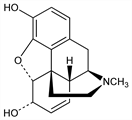
6276. Morphine
Nomenclature
Description and references
Principal alkaloid of opium, q.v.; also found endogenously in various animal tissues including brain, blood and liver. Isoln: F. W. A. Sertürner, Trommsdorff's J. Pharm. 13, 234 (1805); W. Gregory, Ann. 7, 261 (1833). Extraction procedure: L. B. Achor, E. M. K. Geiling, Anal. Chem. 26, 1061 (1954). Structure: J. M. Gulland, R. Robinson, J. Chem. Soc. 123, 980 (1923). Total synthesis: M. Gates, G. Tschudi, J. Am. Chem. Soc. 74, 1109 (1952); 78, 1380 (1956). Biogenesis: E. Leete, J. Am. Chem. Soc. 81, 3948 (1959). Cystallographic study: M. Mackay, D. C. Hodgkin, J. Chem. Soc. 1955, 3261. Configuration: J. Kalvoda et al., Helv. Chim. Acta 38, 1847 (1955). Toxicity data: M. E. Buchwald, G. S. Eadie, J. Pharmacol. Exp. Ther. 71, 197 (1941). GC-MS determn in biological fluids: R. Wasels, F. Belleville, J. Chromatogr. A 674, 225 (1994). Comprehensive description: F. J. Muhtadi, Anal. Profiles Drug Subs. 17, 259-366 (1988). Review of syntheses: T. Hudlicky et al. in Studies in Natural Products Chemistry, Vol. 18, Atta-ur-Rahman, Ed. (Elsevier, Amsterdam, 1996) pp 43-154; J. Zezula, T. Hudlicky, Synlett 2005, 388-405. Review of physiological role of endogenous morphine: G. B. Stefano et al., Trends Neurosci. 23, 436-442 (2000). Review of clinical experience in pain management: S. Donnelly et al., Support. Care Cancer 10, 13-35 (2002); of pharmacokinetics and metabolism in long term treatment: G. Andersen et al., J. Pain Symptom Manage. 25, 74-91 (2003).
Properties
Small rhombic prisms or needles from dil alc as the monohydrate. Becomes anhydrous at 100°, mp 253-254° with decompn. d420 1.32. [α]D25 -132° (methanol). pKb at 20° = 6.13; pKa 9.85. pH of satd soln, 8.5. uv max in acid: 285 nm; in alkali: 298 nm. One gram dissolves in about 5000 ml water, 1100 ml boiling water, 210 ml alcohol, 98 ml boiling alc, 1220 ml chloroform, 6250 ml ether, 114 ml amyl alc, 10 ml boiling methanol, 525 ml ethyl acetate. Freely sol in solns of fixed alkali and alkaline earth hydroxides, in phenol, cresols; moderately sol in mixtures of chloroform with alcohols; slightly sol in ammonia, benzene.Derivative
Hydrochloride.Nomenclature
Properties
Occurs as the trihydrate. White flakes or crystalline powder; bitter taste. Loses its water of crystn at about 100° and usually becomes yellowish. mp about 200° (dec). [α]D25 -113.5° (c = 2.2 in H2O, anhydr basis). One gram dissolves in 17.5 ml water, 0.5 ml boiling water, 52 ml alc, 6 ml alc at 60°; slowly sol in glycerol. Insol in chloroform, ether. pH about 5. Protect from light. LD50 in mice (mg/kg): 226-318 i.v. (Buchwald, Eadie).Derivative
Sulfate Pentahydrate.Nomenclature
Properties
White, fine odorless crystals or powder, or cubical masses. Loses some H2O at ordinary temp; about 3H2O (7.1%) at 100°, the remainder at 130°. Discolors on exposure to light. mp ≈250° with decompn when anhydr. [α]D25 -108.7° (c = 4 in H2O, anhydr basis). pKa 7.9. Partition coefficient (n-octanol/water): 1.42 (at physiological pH). One gram dissolves in 15.5 ml water at 25°, 0.7 ml water at 80°, 565 ml alcohol, 240 ml alcohol at 60°. Insol in chloroform, ether. pH about 4.8. Keep well closed and protected from light.Note
This is a controlled substance (opiate): 21 CFR, 1308.12.Therapeutic Category
Analgesic.
Therapeutic Category (Veterinary)
Analgesic, preanesthetic, antitussive, antiperistaltic.Keywords
Analgesic; Opioids; Phenanthrenes