6286. Moxalactam
Nomenclature
Description and references
Oxa-substituted third generation cephalosporin antibiotic (oxacephalosporin). Prepn: M. Narisada, W. Nagata, DE 2713370; eidem, US 4138486 (1977, 1979 both to Shionogi); M. Narisada et al., J. Med. Chem. 22, 757 (1979). Laboratory evaluation: T. Yoshida et al., Antimicrob. Agents Chemother. 17, 302 (1980). Mechanism of action study: Y. Komatsu, T. Nishikawa, ibid. 316. Pharmacokinetics: R. Wise et al., ibid. 18, 369 (1980). Comparative in vitro activity: T. O. Kurtz et al., ibid. 645. Series of articles on pharmacology, teratology, toxicity studies: Chemotherapy (Tokyo) 28, Suppl. 7, 1002-1235 (1980). Clinical evaluation in neonates and infants: U. B. Schaad et al., J. Pediatr. 98, 129 (1981). Review: Rev. Infect. Dis. 4, Suppl., S489-S726 (1982).
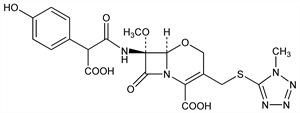
Properties
Colorless powder, mp 117-122° (dec). [α]D25 -15.3 ±2.6° (c = 0.216 in methanol). uv max (methanol): 276 nm (ε 10200).Derivative
Disodium salt.Nomenclature
Properties
[α]D22 -45° (water). uv max (water): 270 nm (ε 12000).Therapeutic Category
Antibacterial.
Keywords
Antibacterial (Antibiotics); β-Lactams; Oxacephems