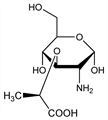
6304. Muramic Acid
Nomenclature
Description and references
Amino sugar found (as the N-acetyl derivative) in peptidoglycan, the main skeletal component of the bacterial cell wall. Discovery: J. T. Park, J. Biol. Chem. 194, 885 (1952). Isoln from spores of Bacillus megatherium: R. E. Strange, F. A. Dark, Nature 177, 186 (1956). Identification and synthesis: R. E. Strange, L. H. Kent, Biochem. J. 71, 333 (1959). Stereospecific synthesis: Y. Matsushima, J. T. Park, J. Org. Chem. 27, 3581 (1962); eidem, Biochem. Prep. 10, 109 (1963); T. Osawa, R. W. Jeanloz, J. Org. Chem. 30, 448 (1965). Review of peptidoglycan structure: H. J. Rogers, Ann. N.Y. Acad. Sci. 235, 29-51 (1974). Use to determine bacterial levels in mammalian tissues: J. Gilbart et al., J. Microbiol. Methods 5, 271 (1986); in airborne dust: A. Fox et al., Appl. Environ. Microbiol. 59, 4354 (1993).