6322. Mycobacidin
Nomenclature
CAS number: 539-35-5
4-Oxo-2-thiazolidinehexanoic acid; ε-[2-(4-thiazolidone)]hexanoic acid; 4-thiazolidone-2-caproic acid; 2-(5-carboxypentyl)-4-thiazolidone; cinnamonin; actithiazic acid; acidomycin. C9H15NO3S; mol wt 217.29.
C 49.75%, H 6.96%, N 6.45%, O 22.09%, S 14.76%.
Description and references
Antibiotic substance produced by Streptomyces lavendulae, Streptomyces virginiae and several other Streptomyces spp. Isoln: Tejera et al., Antibiot. Chemother. 2, 333 (1952). Production by fermentation: Grundy et al., US 2678929 (1954 to Abbott). Synthesis: McLamore et al., J. Am. Chem. Soc. 75, 105 (1953); Nienburg, Friedrichsen, DE 931651 (1955 to BASF). Toxicity studies: Miyake et al., Pharm. Bull. 1, 84, 89 (1953). Review: Caltrider, Antibiotics vol. I, D. Gottlieb, P. D. Shaw, Eds. (Springer-Verlag, New York, 1967) pp 666-668.
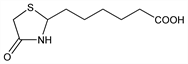
Derivative
l-Form.Properties
Needles from water, ethanol or ethyl acetate, mp 139-140°. [α]D25 -54° (c = 1 in methanol); [α]D25 -60° (c = 1 in ethanol). pK 5.1. Soluble in water, acetone, alcohol, ethylene dichloride, glacial acetic acid. Aq solns are stable over a wide pH range at room temp. Rapidly loses optical activity in dil alkali to give the racemate. Has in vitro activity against Mycobacterium tuberculosis, M. ranae and M. phlei. LD50 in mice (g/kg): 3.5 i.v., 2 s.c. (Miyake).Derivative
Methyl ester.C10H17NO3S; mol wt 231.31.
C 51.92%, H 7.41%, N 6.06%, O 20.75%, S 13.86%.