6326. Mycomycin
Nomenclature
CAS number: 544-51-4
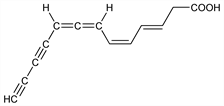
(3E,5Z)-3,5,7,8-Tridecatetraene-10,12-diynoic
acid. C13H10O2; mol wt 198.22.
C 78.77%, H 5.08%, O 16.14%.
Description and references
Acetylenic antibiotic produced by Nocardia acidophilus. Prepn: E. A. Johnson, K. L. Burdon, J. Bacteriol. 54, 281 (1947). Characterization and structure: W. D. Celmer, I. A. Solomons, J. Am. Chem. Soc. 74, 1870, 2245 (1952); 75, 1372 (1953). Synthesis of trans,trans-mycomycins: F. Bohlmann, W. Sucrow, Angew. Chem. 76, 611 (1964).
Properties
Needles from methylene chloride at -40°. Very thermolabile. Complete retention of activity is possible only by storage at -40° or lower. mp 75°. Explosive at melting point. [α]D25 -130° (c = 0.4 in ethanol). uv max (ether): 267, 281 nm (ε 61000, 67000). Sol in alcohol, ether, amyl acetate, methylene chloride. Sparingly sol in water. Relatively stable in extremely dil aq solns (approx 0.005 mg/ml), half-life inversely related to concn. Undergoes an unusual rearrangement in normal aq KOH at 27°, involving an allene to acetylene isomerization accompanied by migration of the existing acetylenic bonds to form isomycomycin.Derivative
Isomycomycin.Nomenclature
CAS number: 505-94-2
3,5-Tridecadien-7,9,11-triynoic acid. C13H10O2; mol wt 198.22.
C 78.77%, H 5.08%, O 16.14%.
CH3‐C≡C‐C≡C‐C≡C‐CH=CHCH=CHCH2COOH. Description and references
Structure and properties: W. D. Celmer, I. A. Solomons, J. Am. Chem. Soc. 74, 3838 (1952).