6327. Mycophenolic Acid
Nomenclature
Description and references
Antibiotic produced by Penicillium brevi-compactum, P. stoloniferum and related spp. Selectively inhibits lymphocyte proliferation by blocking inosine monophosphate dehydrogenase (IMPDH), an enzyme involved in the de novo synthesis of purine nucleotides. Isoln: C. L. Alsberg, O. F. Black, USDA Bur. Plant Ind. Bull. 270, 7 (1912), C.A. 7, 3992 (1913); P. W. Clutterbuck et al., Biochem. J. 26, 1441 (1932); and antimicrobial activity: H. W. Florey et al., Lancet 1, 46 (1946). Structure: J. H. Birkinshaw et al., Biochem. J. 50, 630 (1952); W. R. Logan, G. T. Newbold, J. Chem. Soc. 1957, 1946. Total synthesis: A. J. Birch, J. J. Wright, Aust. J. Chem. 22, 2635 (1969). Biosynthesis: L. Canonica et al., J. Chem. Soc. Perkin Trans. 1 1972, 2639. Fermentation, isoln and biological properties: R. H. Williams et al., Antimicrob. Agents Chemother. 1968, 229. Immunosuppressive effect: A. Mitsui, S. Suzuki, J. Antibiot. 22, 358 (1969). Mechanism of action: A. C. Allison, E. M. Eugui, Transplant. Proc. 26, 3205 (1994); J. T. Ransom, Ther. Drug Monit. 17, 681 (1995). HPLC determn in plasma: J. Shen et al., J. Chromatogr. B 817, 207 (2005); D. Indjova et al., ibid. 327. Evaluation of risk in pregnancy: N. M. Sifontis et al., Transplantation 82, 1698 (2006).
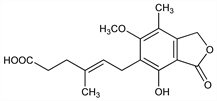
Properties
Needles from hot water, mp 141°. pKa 4.5. Partition coefficient (n-octanol/water): 570 (pH 2); 1.6 (pH 7.4). Almost insol in cold water. Sol in alcohol. LD50 in mice (mg/kg): >1250 orally; 972.9±77 i.p. (Williams).Derivative
2-(4-Morpholinyl)ethyl ester.Nomenclature
Description and references
Prepn: P. H. Nelson et al., US 4753935 (1988 to Syntex); and bioavailability: W. A. Lee et al., Pharm. Res. 7, 161 (1990). Pharmacokinetics: R. E. S. Bullingham et al., Transplant. Proc. 28, 925 (1996). Clinical trial in renal transplantation: P. A. Keown et al., Transplantation 61, 1029 (1996). Reviews: D. O. Taylor et al., J. Heart Lung Transplant. 13, 571-582 (1994); J. J. Lipsky, Lancet 348, 1357-1359 (1996). Review of clinical experience in inflammatory bowel disease: A. C. Ford et al., Aliment. Pharmacol. Ther. 17, 1365-1369 (2003).
Properties
White to off-white crystalline powder, mp 93-94°. pKa 5.6, 8.5. Partition coefficient (n-octanol/water): 0.0085 (pH 2); 238 (pH 7.4). Sol in acetone, methanol; sparingly sol in ethanol. Soly in water (mg/kg): 0.043 (pH 7.4), 4.27 (pH 3.6).Derivative
Sodium salt.Nomenclature
Description and references
Clinical trial in renal transplantation: M. Salvadori et al., Am. J. Transplant. 4, 231 (2003); K. Budde et al., ibid. 237. Review of pharmacology and clinical development of enteric-coated formulation: idem et al., Expert Opin. Pharmacother. 5, 1333-1345 (2004).
Properties
White to off-white, crystalline powder. Highly sol in aqueous media at physiological pH. Practically insol in 0.1N HCl. LD50 in mice (mg/kg): 1176±151 orally; 568±53 i.p. (Williams).Therapeutic Category
Immunosuppressant.
Keywords
IMPDH Inhibitor; Immunosuppressant