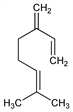
6331. β-Myrcene
Nomenclature
Description and references
Found in oil of bay, verbena, hop, and others. Isoln: Power, Kleber, Pharm. Rundsch. 13, 60 (1895); see also Ruzicka, Stoll, Helv. Chim. Acta 7, 272 (1924); Goulding, Roberts, J. Chem. Soc. 105, 2614 (1914); Booker et al., ibid. 1940, 1453; Kugler, Kováts, Helv. Chim. Acta 46, 1480 (1963). Obtained by pyrolysis of β-pinene: Goldblatt, Palkin, J. Am. Chem. Soc. 63, 3517 (1941); Rummelsburg, US 2444790 (1948 to Hercules Powder); Houlihan et al., J. Am. Chem. Soc. 81, 4692 (1959). Separation of isomers: Ohloff et al., Ann. 675, 83 (1964). Synthesis: O. P. Vig et al., Indian J. Chem. 7, 450 (1969); 11, 104 (1973); 13, 1244 (1975); T. Mandai et al., Tetrahedron Lett. 22, 763 (1981). Formation from isoprene: K. Takabe et al., Synthesis 1977, 307.