6332. Myricetin
Nomenclature
CAS number: 529-44-2
3,5,7-Trihydroxy-2-(3,4,5-trihydroxyphenyl)-4H-1-benzopyran-4-one; 3,3′,4′,5,5′,7-hexahydroxyflavone; cannabiscetin; delphidenolon 1575. C15H10O8; mol wt 318.24.
C 56.61%, H 3.17%, O 40.22%.
Description and references
From the bark of Myrica nagi Thunb., Myriaceae: Perkin, Hummel, J. Chem. Soc. 69, 1287 (1896). Structure: Perkin, ibid. 81, 203 (1902). Identity with cannabiscetin: Seshadri, Venkateswarlu, Proc. Indian Acad. Sci. 23A, 296 (1946); C.A. 40, 6447 (1946). Synthesis: Kalff, Robinson, J. Chem. Soc. 127, 181 (1925); Rao et al., J. Sci. Ind. Res. 8B, No. 6, 113 (1949). Occurrence in Hamamelidaceae and Anacardiaceae: Reznek, Egger, Z. Naturforsch. 15b, 247 (1960). Metabolism: Smith, Griffiths, Biochem. J. 118, 53p (1970).
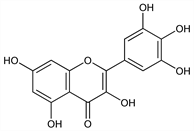
Properties
Yellow needles from dil alc, mp 357°. uv max (ethanol): 375, 255 nm. Sparingly sol in boiling water; sol in alcohol. Practically insol in chloroform, acetic acid.Derivative
Hexaacetate.C27H22O14; mol wt 570.46.
C 56.85%, H 3.89%, O 39.27%.
Properties
Crystals, mp 213°.Derivative
Hexaethyl ether.C27H34O8; mol wt 486.55.
C 66.65%, H 7.04%, O 26.31%.
Properties
Needles from alcohol, mp 149-151°.Derivative
3-Rhamnoside.Nomenclature
Myricitrin.C21H20O12; mol wt 464.38.
C 54.31%, H 4.34%, O 41.34%.