6363. Naltrexone
Nomenclature
Description and references
Nonselective opioid receptor antagonist; congener of naloxone, q.v. Prepn: H. Blumberg et al., US 3332950 (1967 to Endo). Metabolism study: E. J. Cone, Tetrahedron Lett. 1973, 2607. Pharmacology and toxicity: R. P. Maickel et al., Ann. N.Y. Acad. Sci. 281, 321 (1976). Clinical pharmacology: E. R. Gritz et al., Clin. Pharmacol. Ther. 19, 773 (1976); K. Verebey et al., ibid. 20, 315 (1976). GC determn in urine: H. E. Dayton, C. E. Inturrisi, Drug Metab. Dispos. 4, 474 (1976). LC-MS/MS determn in plasma: H. Yun et al., Talanta 71, 1553 (2007). Review of pharmacokinetics and therapeutic use in opioid dependence: J. P. Gonzales, R. N. Brogden, Drugs 35, 192-213 (1988). Clinical trials in alcoholism: B. J. Berg et al., Drug Saf. 15, 274 (1996); R. S. Croop et al., Arch. Gen. Psychiatry 54, 1130 (1997). Review of clinical experience in alcoholism: H. M. Pettinati et al., J. Clin. Psychopharmacol. 26, 610-625 (2006); of depot preparations: B. A. Johnson, Expert Opin. Pharmacother. 7, 1065-1073 (2006).
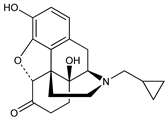
Properties
Crystals from acetone, mp 168-170°. Sol in ethanol. Insol in water. LD50 in mice (mg/kg): 586 s.c. (Maickel).Derivative
Hydrochloride.Nomenclature
Properties
Crystals from methanol, mp 274-276°.Therapeutic Category
Narcotic antagonist. In treatment of alcoholism.
Therapeutic Category (Veterinary)
Adjunct in treatment of behavioral disorders.Keywords
Narcotic Antagonist; Alcohol Dependence Treatment