6362. Naloxone
Nomenclature
Description and references
Specific opioid antagonist. Prepn: GB 939287 (1963 to Sankyo); M. J. Lewenstein, J. Fishman, US 3254088 (1966); R. A. Olofson et al., Tetrahedron Lett. 1977, 1567. Clinical pharmacology and abuse potential: D. R. Jasinski et al., J. Pharmacol. Exp. Ther. 157, 420 (1967). Metabolism: J. M. Fujimoto, Proc. Soc. Exp. Biol. Med. 133, 317 (1970). Synthesis of (+)-form and stereospecific activity: I. Iijima et al., J. Med. Chem. 21, 398 (1978). Clinical trial in Alzheimer's disease: B. Reisberg et al., N. Engl. J. Med. 308, 721 (1983). Comprehensive description: M. M. A. Hasson et al., Anal. Profiles Drug Subs. 14, 453-489 (1985). Review of pharmacology and therapeutic uses: L. F. McNicholas, W. R. Martin, Drugs 27, 81-93 (1984); of clinical experience in Alzheimer's disease: C. Stowe, M. L. Gora, Ann. Pharmacother. 27, 447-448 (1993); in drug overdose: J. M. Chamberlain, B. L. Klein, Am. J. Emerg. Med. 12, 650-660 (1994).
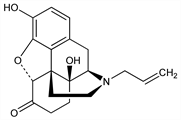
Properties
Crystals from ethyl acetate, mp 184° (Lewenstein), 177-178° (Sankyo Co.). [α]D20 -194.5° (c = 0.93 in CHCl3). Sol in chloroform. Practically insol in petr ether.Derivative
Hydrochloride.Nomenclature
Properties
Crystals from ethanol + ether, mp 200-205°. Sol in water, alcohol. Practically insol in ether.Therapeutic Category
Narcotic antagonist.
Therapeutic Category (Veterinary)
Narcotic antagonist.Keywords
Narcotic Antagonist