6368. Naphazoline
Nomenclature
CAS number: 835-31-4
4,5-Dihydro-2-(1-naphthalenylmethyl)-1H-imidazole; 2-(1-naphthylmethyl)imidazoline. C14H14N2; mol wt 210.27.
C 79.97%, H 6.71%, N 13.32%.
Description and references
α-Adrenergic agonist. Prepd by reacting the acetic acid anhydride of naphthoimidic acid with ethylenediamine: A. Sonn, US 2161938 (1939 to Soc. Chem. Ind. Basle); by reacting naphthylthioacetamide with ethylenediamine: DK 62889 (1944 to Lovens Kemiske), C.A. 40, 4398 (1946). Toxicity data: J. Gylfe et al., Fed. Proc. 9, 280 (1950). Comprehensive description: G. M. Wall, Anal. Profiles Drug Subs. Excip. 21, 307-344 (1992).
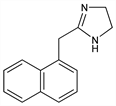
Derivative
Hydrochloride.Nomenclature
CAS number: 550-99-2
Ak-Con (Akorn); Albalon (Allergan); Clera (Person & Covey); Coldan; Iridina Due (Montefarmaco); Naphcon (Alcon); Niazol; Opcon (Bausch & Lomb); Privine (Novartis); Rhinantin; Rhinoperd; Sanorin; Sanorin-Spofa; Strictylon. C14H14N2.HCl; mol wt 246.74.
C 68.15%, H 6.13%, N 11.35%, Cl 14.37%.
Properties
Bitter crystals, mp 255-260°. uv max (ethanol): 223, 270, 280, 287, 291 nm (E1%1cm 3622, 239, 286, 196, 198). pKa (25°C) 10.35 ±0.02, (35°C) 10.13 ±0.02, (45°C) 9.92 ±0.03. pKa (25°C) 10.35 ±0.02, (35°C) 10.13 ±0.02, (45°C) 9.92 ±0.03. Freely sol in water (40 g dissolve in 100 ml) and in alcohol. Slightly sol in chloroform. Insol in benzene, ether. A 1% aq soln has a pH of ≈6.2. LD50 s.c. in rats: 385 mg/kg (Gylfe).Therapeutic Category
Adrenergic (vasoconstrictor); decongestant.
Keywords
α-Adrenergic Agonist; Decongestant