6422. Narceine
Nomenclature
CAS number: 131-28-2
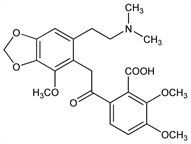
6-[[6-[2-(Dimethylamino)ethyl]-4-methoxy-1,3-benzodioxol-5-yl]acetyl]-2,3-dimethoxybenzoic
acid; 6-[[6-[2-(dimethylamino)ethyl]-2-methoxy-3,4-(methylenedioxy)phenyl]acetyl]-o-veratric acid. C23H27NO8; mol wt 445.46.
C 62.01%, H 6.11%, N 3.14%, O 28.73%.
Description and references
Opioid analgesic. Occurs in opium to the extent of 0.1-0.5%. The separation from morphine mother liquors is tedious: Merck, Chem. Ztg. 13, 525 (1889). Prepn from narcotine or gnoscopine: Roser, Ann. 247, 167 (1888); Frankforter, Keller, Ann. Chem. J. 22, 61 (1899); Frerichs, Arch. Pharm. 241, 259 (1903); Hope, Robinson, J. Chem. Soc. 105, 2100 (1914). Structure: Freund, Frankforter, Ann. 277, 20 (1893); Freund, Michaels, Ann. 286, 248 (1895); Freund, Ber. 40, 194 (1907); Freund, Oppenheim, Ber. 42, 1084 (1909); Addinall, Major, J. Am. Chem. Soc. 55, 1202, 2153 (1933).
Properties
The anhydr material is very hygroscopic, mp 138°. uv max (ethanol): 270 nm (log ε 3.98). Usually the alkaloid is obtained as the trihydrate. Clusters of silky, prismatic needles from water, mp 176°. pKb (20°) = 10.7, pKa = 9.3. pH of satd soln = 5.8. One gram dissolves in 770 ml water, 220 ml boiling water. Moderately sol in hot alcohol, nearly insol in benzene, chloroform, ether, petr ether; sol in alkali hydroxide solns forming salts, also in dil mineral acids.Derivative
Ethylnarceine hydrochloride.Nomenclature
Narcyl.C25H32ClNO8; mol wt 509.98.
C 58.88%, H 6.32%, Cl 6.95%, N 2.75%, O 25.10%.
Properties
Plates from water, mp 208-210°. Slightly sol in cold water, sol in hot water, alcohol, chloroform; insol in ether.Therapeutic Category
Antitussive.
Keywords
Antitussive