6438. Nefazodone
Nomenclature
CAS number: 83366-66-9
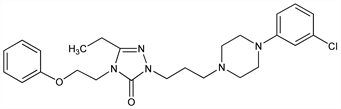
2-[3-[4-(3-Chlorophenyl)-1-piperazinyl]propyl]-5-ethyl-2,4-dihydro-4-(2-phenoxyethyl)-3H-1,2,4-triazol-3-one; 2-[3-[4-(3-chlorophenyl)-1-piperazinyl]propyl]-5-ethyl-4-(2-phenoxyethyl)-2H-1,2,4-triazol-3(4H)-one. C25H32ClN5O2; mol wt 470.01.
C 63.89%, H 6.86%, Cl 7.54%, N 14.90%, O 6.81%.
Description and references
Selective serotonin 5-HT2 receptor antagonist. Prepn: D. L. Temple, Jr., W. G. Lobeck, Jr., US 4338317 (1982 to Mead Johnson). Synthesis and x-ray crystal structure: G. D. Madding et al, J. Heterocycl. Chem. 22, 1121 (1985). Pharmacology: A. S. Eison et al., Psychopharmacol. Bull. 26, 311 (1990). HPLC determn in plasma: J. E. Franc et al., J. Chromatogr. 570, 129 (1991). Clinical trial in depression: M. F. D'Amico et al., Psychopharmacol. Bull. 26, 147 (1990); in combination with psychotherapy for chronic depression: M. B. Keller et al., N. Engl. J. Med. 342, 1462 (2000). Review: W. E. Heydorn, Expert Opin. Invest. Drugs 4, 131-137 (1995).
Properties
Crystals from 2-propanol/heptane, mp 83-84°.Derivative
Hydrochloride.Nomenclature
CAS number: 82752-99-6
BMY-13754; MJ-13754-1; Dutonin (BMS); Serzone (BMS). C25H32ClN5O2.HCl; mol wt 506.47.
C 59.29%, H 6.57%, Cl 14.00%, N 13.83%, O 6.32%.
Properties
Crystals from 2-propanol, with slow cooling yields a polymorph, mp 186.0-187.0°; with rapid cooling yields a polymorph, mp 181.0-182.0° (Madding). Also reported as crystals from ethanol, mp 175-177° (Temple).Therapeutic Category
Antidepressant.
Keywords
Antidepressant; Serotonin Receptor Antagonist