6454. Neomycin
Nomenclature
Description and references
Antibiotic complex composed of neomycins A, B and C. Produced by Streptomyces fradiae: Waksman, Lechevalier, Science 109, 305 (1949); Waksman et al., J. Clin. Invest. 28, 934 (1949); Swart et al., ibid. 1045; Waksman, Lechevalier, US 2799620 (1957). Purification: Jackson, US 2848365 (1958 to Upjohn); Haak, US 3108996 (1963 to Upjohn). Recovery: Miller, US 3005815 (1961 to Merck & Co.); Moses, US 3022228 (1962 to Penick); GB 945475 (1964 to O.W.G. Chemie). Characterization: Peck et al., J. Am. Chem. Soc. 71, 2590 (1949); Regna, Murphy, ibid. 72, 1045 (1950); Dutcher et al., ibid. 73, 1384 (1951). Structure of neomycins B and C: K. L. Rinehart et al., ibid. 79, 4567, 4568 (1957); ibid. 84, 3216, 3218 (1962). Abs config of neomycin C: M. Hichens, K. L. Rinehart, ibid. 85, 1547 (1963). Total synthesis of neomycin C: S. Umewaza, Y. Nishimura, J. Antibiot. 30, 189 (1977); S. Umewaza et al., Bull. Chem. Soc. Jpn. 53, 3259 (1980); of neomycin B: T. Usui, S. Umezawa, J. Antibiot. 40, 1464 (1987). Monographs: S. A. Waksman, Neomycin (Rutgers Univ. Press, New Brunswick, N. J., 1953) 219 pp; K. L. Rinehart, Jr., The Neomycins and Related Antibiotics (Wiley, New York, 1964) 137 pp. Comprehensive description: W. F. Heyes, Anal. Profiles Drug Subs. 8, 399-488 (1979).
Properties
Neomycin complex is an amorphous base sol in water, methanol and acidified alcohol. Practically insol in common organic solvents. Solns up to 250 mg/ml H2O may be prepared.Derivative
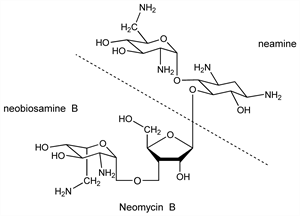
Neomycin A see Neamine.Derivative
Neomycin B.Nomenclature
Properties
Identity of neomycin B and framycetin: K. L. Rinehart et al., ibid. 82, 3938 (1960). Yields on hydrolysis neamine and neobiosamine B. Structure of neobiosamine B: K. L. Rinehart et al., J. Am. Chem. Soc. 82, 2970 (1960).Derivative
Neomycin B hydrochloride.Nomenclature
Properties
Amorphous white powder. [α]D20 +57° (H2O). Soly in mg/ml at ≈28°: water 15.0; methanol 5.7; ethanol 0.65; isopropanol 0.05; isoamyl alcohol 0.33; cyclohexane 0.06; benzene 0.03. Practically insol in acetone, ether, other organic solvents. For additional soly data see Weiss et al., Antibiot. Chemother. 7, 374 (1957).Derivative
Neomycin B sulfate.Nomenclature
Properties
Amorphous white powder. Practically tasteless. [α]D20 +54° (c = 2 in H2O). Soly in mg/ml at ≈28°: water 6.3; methanol 0.225; ethanol 0.095; isopropanol 0.082; isoamyl alcohol, 0.247; cyclohexane 0.08; benzene 0.05. Practically insol in acetone, ether, chloroform. Aq solns are fairly stable at pH 2 to 9. Highly purified prepns are very stable to alkali and unstable to acids. Refluxing with barium hydroxide for 18 hrs showed no loss of activity. Boiling with mineral acids yields an aldehyde, characterized as furfural, and an organic base.Derivative
Neomycin C.Nomenclature
Properties
Yields on hydrolysis neamine and neobiosamine C. Structure of neobiosamine C: K. L. Rinehart, P. Woo, J. Am. Chem. Soc. 80, 6463 (1958).Therapeutic Category
Antibacterial.
Therapeutic Category (Veterinary)
Antibacterial.Keywords
Antibacterial (Antibiotics); Aminoglycosides