6531. Niflumic Acid
Nomenclature
CAS number: 4394-00-7
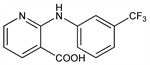
2-[[3-(Trifluoromethyl)phenyl]amino]-3-pyridinecarboxylic acid; 2-(α,α,α-trifluoro-m-toluidino)nicotinic acid; 2-[3-(trifluoromethyl)anilino]nicotinic acid; UP-83; Actol (Fournier); Forenol (Roemmers); Landruma (Landerln); Nifluril. C13H9F3N2O2; mol wt 282.22.
C 55.33%, H 3.21%, F 20.20%, N 9.93%, O 11.34%.
Description and references
Prepn: NL 6414717; C. Hoffmann, A. Faure, US 3415834 (1965, 1968 both to Lab. U.P.S.A.); eidem, Bull. Soc. Chim. Fr. 1966, 2316. Pharmacological and metabolic studies: Glasson et al., Biochem. Pharmacol. 18, 633 (1969); Boissier et al., Therapie 26, 211 (1971). Toxicity data: J. R. J. Sorenson, J. Med. Chem. 19, 135 (1976). Determn in human plasma by GLC: G. Houin et al., J. Chromatogr. 223, 351 (1981).
Properties
Crystals from ethanol, mp 204°. LD50 in rats (mg/kg): 370 orally; 155 i.p. (Sorenson).Derivative
β-Morpholinoethyl ester.Nomenclature
CAS number: 65847-85-0
Morniflumate; Flomax (Chiesi); Nifluril (suppositories) (UPSA). C19H20F3N3O3; mol wt 395.38.
C 57.72%, H 5.10%, F 14.42%, N 10.63%, O 12.14%.
Description and references
Prepn of the ester HCl: Hoffmann, DE 1802777; idem, US 3708481 (1969, 1973 both to Hexachimie).
Derivative
Phthalidyl ester see Talniflumate.Therapeutic Category
Anti-inflammatory.
Keywords
Anti-inflammatory (Nonsteroidal); Aminoarylcarboxylic Acid Derivatives