6661. Nizofenone
Nomenclature
CAS number: 54533-85-6
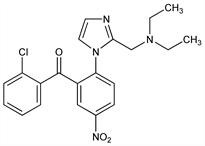
(2-Chlorophenyl)[2-[2-[(diethylamino)methyl]-1H-imidazol-1-yl]-5-nitrophenyl]methanone; 2′-chloro-2-[2-[(diethylamino)methyl]imidazol-1-yl]-5-nitrobenzophenone; 1-[2-(2-chlorobenzoyl)-4-nitrophenyl]-2-(diethylaminomethyl)imidazole. C21H21ClN4O3; mol wt 412.87.
C 61.09%, H 5.13%, Cl 8.59%, N 13.57%, O 11.63%.
Description and references
Imidazole derivative exhibiting protective activity against cerebral anoxia or ischemia. Prepn: M. Nakanishi et al., DE 2403416; eidem, US 3915981 (1974, 1975 both to Yoshitomi). Antianoxic effect in animal models: H. Yasuda et al., Arch. Int. Pharmacodyn. 233, 136 (1978). Mechanism of action: eidem, ibid. 242, 77 (1979). Multicenter clinical studies: I. Saito et al., Neurol. Res. 5, 29 (1983); T. Ohta et al., J. Neurosurg. 64, 420 (1986). Toxicity studies: H. Horizoe et al., Oyo Yakuri 30, 627 (1985), C.A. 104, 61754m (1986); K. Okumura et al., ibid. 633, C.A. 104, 61755 (1986).
Properties
Pale yellow crystals from isopropyl ether, mp 75-76°.Derivative
Fumarate.Nomenclature
CAS number: 54533-86-7
Midafenone; Y-9179; Ekonal (Yoshitomi). C21H21ClN4O3.C4H4O4; mol wt 528.94.
C 56.77%, H 4.76%, Cl 6.70%, N 10.59%, O 21.17%.
Properties
Pale yellow crystals from isopropyl ether, mp 157-158°. LD50 in male, female mice, male, female rats (mg/kg): 495, 504, 1711, 1580 orally; 62, 70, 63, 65 i.v.; 270, 278, 1830, 1629 s.c. (Horizoe).Therapeutic Category
Nootropic.
Keywords
Nootropic