6692. Nordefrin
Nomenclature
CAS number: 6539-57-7 (unspecified stereo);
74812-63-8
(R*,S*-(±)-form)
4-(2-Amino-1-hydroxypropyl)-1,2-benzenediol; α-(1-aminoethyl)-3,4-dihydroxybenzyl alcohol; 3,4-dihydroxynorephedrine; 3,4-dihydroxyphenylpropanolamine; 3,4-dihydroxyphenylaminopropanol; α-(α-aminoethyl)protocatechuyl alcohol; α-methylnoradrenaline; α-methylnorepinephrine; norhomoepinephrine; isoadrenaline; aminopropanolpyrocatechol; homoarterenol. C9H13NO3; mol wt 183.20.
C 59.00%, H 7.15%, N 7.65%, O 26.20%.
Description and references
α-Adrenergic agonist. Prepn: DE 254438 (1912 to Bayer); W. H. Hartung et al., J. Am. Chem. Soc. 53, 4149 (1931); M. Bockmühl et al., US 1948162 (1934 to Winthrop); Bruckner et al., Ber. 76B, 466 (1943). Configuration: Fodor et al., Monatsh. Chem. 83, 1146 (1952). Prepn of optical isomers: DE 269327 (1913 to Bayer); Bockmühl, Gorr, DE 639126 (1936 to I. G. Farbenind.). Pharmacology: F. P. Luduena et al., J. Dent. Res. 37, 206 (1958). HPLC determn in plasma: D. A. Jenner et al., J. Chromatogr. 224, 507 (1981). Effect on toxicity of local anesthetics: S. E. Taylor, R. L. Dorris, Anesth. Prog. 36, 79 (1989). Review of use in local anesthesia: T. M. Hensley et al., J. Foot Surg. 26, 504-510 (1987).
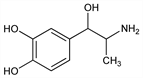
Derivative
Hydrochloride.Nomenclature
CAS number: 138-61-4 (unspecified stereo);
61-96-1
(R*,S*-(±)-form)
Corbasil; Cobefrin (Winthrop). C9H13NO3.HCl; mol wt 219.67.
C 49.21%, H 6.42%, N 6.38%, O 21.85%, Cl 16.14%.
Properties
Crystals, dec 178-179°. One gram dissolves in about 1.5 ml water, 15 ml alcohol. Practically insol in ether. Solns are neutral and are easily destroyed by traces of alkali.Derivative
(-)-Form.Nomenclature
CAS number: 829-74-3
Corbadrine; levonordefrin; Neo-Cobefrin (Cook-Waite). Properties
Crystals, dec 212-215°. [α]D25 -31.0° (c = 0.5 in 0.01N HCl). uv max (dil HCl): 278 nm. Practically insol in water. Freely sol in aq solns of mineral acids. Very slightly sol in acetone, chloroform, ethanol, ether. LD50 in mice (mg/kg): 12.6 i.v. (Luduena).Therapeutic Category
Vasoconstrictor.
Keywords
Decongestant