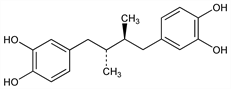
6693. Nordihydroguaiaretic Acid
Nomenclature
Description and references
Lipoxygenase inhibitor; occurs as the meso-form in the resinous exudate of the creosote bush, Larrea divaricata, Zygophyllaceae (Covillea tridentata). Isoln: C. W. Waller, O. Gisvold, J. Am. Pharm. Assoc. 34, 78 (1945). Prepn from guaiaretic acid dimethyl ether: G. Schroeter et al., Ber. 51, 1587 (1918); R. D. Haworth et al., J. Chem. Soc. 1934, 1423. Synthesis: S. V. Lieberman et al., J. Am. Chem. Soc. 69, 1540 (1947); and configuration of naturally occurring form: C. W. Perry et al., J. Org. Chem. 37, 4371 (1972). Use as antioxidant: W. M. Lauer, US 2373192 (1945 to U. S. Secr'y of Agriculture). Antiproliferative effect on cultured keratinocytes: D. I. Wilkinson, E. K. Orenberg, Int. J. Dermatol. 26, 660 (1987); on cultured glioma cells: D. E. Wilson et al., J. Neurosurg. 71, 551 (1989). Clinical trial in actinic keratoses: E. A. Olsen et al., J. Am. Acad. Dermatol. 24, 738 (1991).
Properties
Crystals from acetic acid, mp 185-186°. uv max (methanol): 283, 218 nm (ε 6660, 13400). Sol in ethanol, methanol, ether, concd H2SO4; slightly sol in hot water, chloroform; sol in dil alkalies, developing a deep red color. Practically insol in petr ether, benzene, toluene, dil HCl.Use
As antioxidant for fats and oils in foods.Therapeutic Category
Antineoplastic.