6707. Norlevorphanol
Nomenclature
CAS number: 1531-12-0
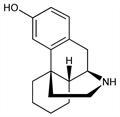
Morphinan-3-ol; (-)-3-hydroxymorphinan; (-)-1,3,4,9,10,10a-hexahydro-6-hydroxy-2H-10,4a-iminoethanophenanthrene; NIH-7539. C16H21NO; mol wt 243.34.
C 78.97%, H 8.70%, N 5.76%, O 6.57%.
Description and references
Opioid analgesic. Prepn of (-) and (+) isomers: O. Schnider, A. Grüssner, Helv. Chim. Acta 34, 2211 (1951); J. Hellerbach, et al., ibid. 39, 429 (1956); GB 765920 (1957 to Hoffmann-La Roche).
Properties
Crystals from acetone + methanol, mp 270-272°. [α]D21 -42 ±2° (c = 1 in methanol).Derivative
Hydrobromide.Nomenclature
CAS number: 63732-85-4
C16H21NO.HBr; mol wt 324.26.
C 59.26%, H 6.84%, N 4.32%, O 4.93%, Br 24.64%.
Properties
Crystals from water, mp 222-224°.Derivative
(+)-Form.Nomenclature
CAS number: 15676-23-0
(9α,13α,14α)-Morphinan-3-ol; nordextrorphan. Description and references
Secondary metabolite of dextromethorphan, q.v.: G. Pfaff et al., Int. J. Pharm. 14, 173 (1983). Stereocontrolled synthesis: Y. Génisson et al., J. Org. Chem. 58, 2052 (1993).