6716. Norvaline
Nomenclature
CAS number: 6600-40-4
l-Norvaline; (S)-2-Aminovaleric acid; l-α-aminovaleric acid; (S)-2-aminopentanoic acid. C5H11NO2; mol wt 117.15.
C 51.26%, H 9.46%, N 11.96%, O 27.31%.
Description and references
Prepd by treating butyraldehyde ammonia with HCN and HCl: Slimmer, Ber. 35, 404 (1902); from 2-acetylvaleric acid ethyl ester: Hamlin, Hartung, J. Biol. Chem. 145, 349 (1942); from acetamidomalonic acid diethyl ester: Archer, Albertson, US 2445817 (1948 to Winthrop-Stearns); from 1-nitrobutane: Stiles, Finkbeiner, J. Am. Chem. Soc. 85, 616 (1963); US 3055936 (1962 to Res. Corp.). Prepn of optically active forms: Abderhalden, Kurton, Fermentf. 4, 328; Chem. Zentralbl. 1921 III,, 296.
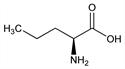
Properties
Crystals from dil alc. mp ≈305° (closed capillary). [M]D +29.2° (5N HCl); [M]D +41.0° (glacial acetic acid). [α]D20 +23.0° (c = 10 in 20% HCl). Freely sol in hot water; insol in alcohol, ether, chloroform, ethyl acetate, petr ether.Derivative
dl-Form.Nomenclature
CAS number: 760-78-1
Properties
Minute leaflets from alcohol or water, mp 303° (closed capillary). pK1′ 2.36; pK2′ 9.72. Sublimes without decompn. One gram dissolves in 10 ml water at 18°. Freely sol in hot water; practically insol in alcohol, ether, chloroform, ethyl acetate, petr ether.Derivative
d(-)-Form.Nomenclature
CAS number: 2013-12-9